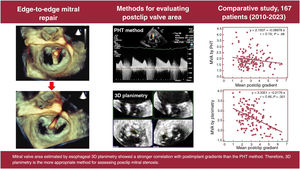
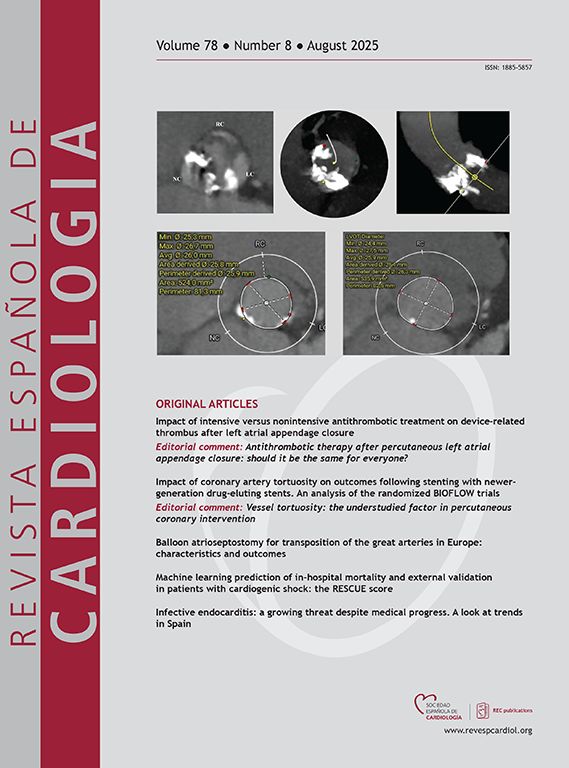
There is limited evidence to identify the most accurate method for measuring the mitral valve area (MVA) after percutaneous edge-to-edge mitral repair. Our objective was to evaluate the optimal method in this context and its correlation with the mean transmitral gradient.
MethodsA registry of patients undergoing percutaneous mitral repair was conducted, analyzing different methods of measuring MVA and their correlation with the mean gradient.
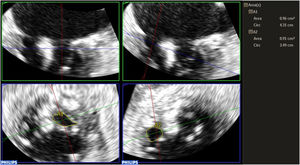
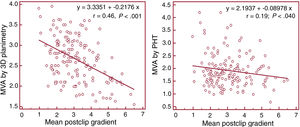
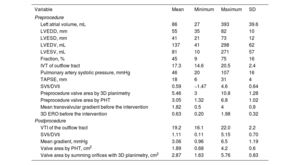
ResultsWe analyzed data from 167 patients. The mean age was 76±10.3 years, 54% were men, and 46% were women. Etiology was degenerative in 45%, functional in 39%, and mixed in 16%. Postclip MVA measurements were 1.89±0.60 cm2 using pressure half-time (PHT), 2.87±0.83 cm2 using 3D planimetry, and the mean gradient was 3±1.19mmHg. MVA using 3D planimetry showed a stronger correlation with the mean gradient (r=0.46, P<.001) than MVA obtained by PHT (r=0.19, P=.048). Interobserver agreement was also higher with 3D planimetry than with PHT (intraclass correlation coefficient of 0.90 vs 0.81 and variation coefficient of 9.6 vs 19.7%, respectively).
ConclusionsOur study demonstrates that the PHT method significantly underestimates MVA after clip implantation compared with direct measurement using transesophageal 3D planimetry. The latter method also correlates better with postimplantation gradients and has less interobserver variability. These results suggest that 3D planimetry is a more appropriate method for assessing postclip mitral stenosis.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm