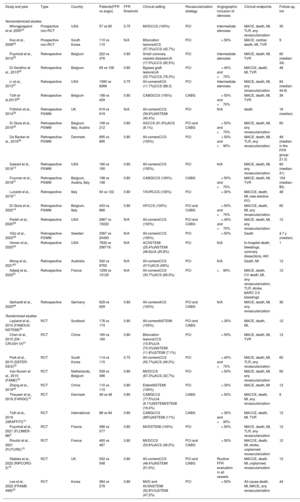
Several studies have investigated the effectiveness of fractional flow reserve (FFR) guidance in improving clinical outcomes after myocardial revascularization, yielding conflicting results. The aim of this study was to compare clinical outcomes in patients with coronary artery disease following FFR-guided or angiography-guided revascularization.
MethodsBoth randomized controlled trials (RCTs) and nonrandomized intervention studies were included. Coprimary endpoints were all-cause death, myocardial infarction, and major adverse cardiovascular events (MACE). The study is registered with PROSPERO (CRD42022344765).
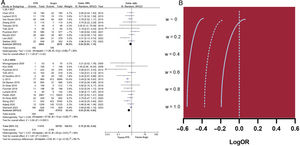
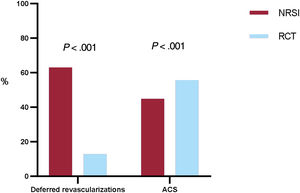
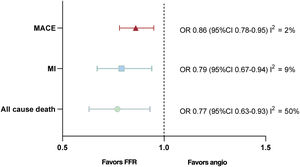
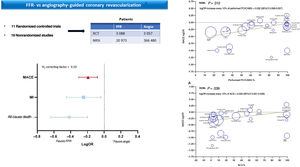
ResultsA total of 30 studies enrolling 393 588 patients were included. FFR-guided revascularization was associated with significantly lower rates of all-cause death (OR, 0.63; 95%CI, 0.53-0.73), myocardial infarction (OR, 0.70; 95%CI, 0.59-0.84), and MACE (OR, 0.77; 95%CI, 0.70-0.85). When only RCTs were considered, no significant difference between the 2 strategies was observed for any endpoints. However, the use of FFR was associated with reduced rates of revascularizations and treated lesions. Metaregression suggested that the higher the rate of revascularized patients the lower the benefit of FFR guidance on MACE reduction compared with angiography guidance (P=.012). Similarly, higher rates of patients with acute coronary syndromes were associated with a lower benefit of FFR-guided revascularization (P=.039).
ConclusionsFFR-guided revascularization was associated with lower rates of all-cause death, myocardial infarction and MACE compared with angiographic guidance, with RCTs and nonrandomized intervention studies yielding conflicting data. The benefits of FFR-guidance seem to be less evident in studies with high revascularization rates and with a high prevalence of patients with acute coronary syndrome.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm