Pulmonary hypertension (PH) is an irreversible progressive disease that, unless diagnosed early, can lead to right ventricular failure and end-stage heart failure (HF). Significant advances have been made in pharmacological treatment,1 but life expectancy remains short—57% at 5 years—and the disease is more aggressive in children than in adults.2
Atrial septostomy (AS) is indicated in children with PH in functional class III or IV of the Ross classification who experience recurrent syncope and/or right HF refractory to medical treatment, as a bridge to lung transplant.3,4 The main contraindications are a mean atrial pressure > 20 mmHg and oxygen saturation <90%.5
AS can be performed using a number of techniques; one of the more commonly used, and which is the practice in our hospital, involves placing a stent in the interatrial septum. This technique is performed with a 7-Fr sheath and a pediatric Brockenbrough needle, which passes first through the inferior vena cava in a 15-mm snare that is positioned from the contralateral vein. Once the right atrial pressure has been checked to ensure it does not contraindicate the procedure, the septum is punctured, the sheath is positioned in the left atrium, and a coronary guidewire is advanced to the pulmonary vein. A 10×19mm Palmaz Genesis large stent is positioned in the center of the septum. The snare is then positioned halfway along the stent, and the stent is dilatated, restricted in the middle by the snare so that it takes on an hourglass shape and becomes highly stable. Balloon dilatation is then performed with coronary or peripheral balloons, based on saturation levels, up to a maximum of 5-7mm depending on the size of the patient. The size of the defect can be reduced again if deemed necessary, by tightening the snare, so that the defect can be modified and adapted to the individual patient.
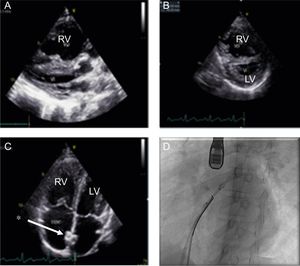
We retrospectively reviewed the clinical histories of all patients of pediatric age with a diagnosis of PH who underwent AS at our center: 90.9% of the procedures performed in the last 10 years. The procedure was performed under general anesthetic and with transesophageal ultrasound guidance (Figure 1), and as part of the protocol patients had a 24-hour observation period in the pediatric intensive care unit with standard care pulmonary vasodilators.
A: echocardiography, showing RV dilatation compressing the LV. B: type IV IVS and abnormal sphericity index, echocardiographic signs of severe PH. C: image of a correctly positioned stent (*). D: catheterization, positioning a stent with TEE guidance. IVS, interventricular septum; LV, left ventricle; RV, right ventricle; TEE, transesophageal echocardiography.
Statistical analysis was performed with SPSS STATISTICS Version 24 (IBM Corp). Continuous variables were analyzed as mean (range), and categorical variables, as percentages. Symptom resolution was analyzed using the chi-square test (Fisher exact test), and P <.05 was considered statistically significant. Kaplan-Meier survival curves were plotted.
AS was performed in 11 of the 45 children under follow-up for PH (24.4%), all of whom had type I PH (idiopathic or familiar in 8 of them); sex distribution was even (45.5% were girls). The mean age at diagnosis and at the time of AS was 3.10 (0.08-7.90) and 4.86 (0.39-11.87) years, respectively. The mean time between diagnosis of PH and the procedure was 20.4 months (Table 1).
Patient Data and Procedures for Atrial Septostomy in Pediatric Age
| Age at Dx, years | Age at AS, years | Sex | Type of PH I | Indication for AS | PVD treatment | Type of AS | AS, mm | RAP, mmHg | Mean PAP, mmHg | Sa O2pre-AS, mmHg | Sa O2post-AS, mmHg | TAPSE pre-AS, mm | TAPSE post-AS, mm | Complications | Outcome | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 3.4 | 8.75 | M | CHD | RRHF | Bosentan, sildenafil, iloprost | Stent | 5 | 11 | 46 | 96 | 100 | 12 | 12 | No | Initially functional class III then class II |
| 2 | 2.8 | 2.85 | M | Idiopathic | RRHF | Nitric oxide, sildenafil, prostacyclin | Stent | 10 | 8 | 81 | 100 | 95 | 13 | 12 | No | No improvement in functional classDied 34 days after AS |
| 3 | 7.2 | 7 | F | Idiopathic | RRHF | Nitric oxide | Angioplasty and balloon dilatation | 12 | 8 | 62 | 97 | 97 | 17 | 17 | No | Initially class IV then class II |
| 4 | 7.9 | 11.8 | F | Idiopathic | ES | Bosentan, sildenafil, iloprost | Stent | 4 | 8 | 77 | 100 | 91 | 13 | 16 | No | No further syncopeTransplantation at 6.5 months after AS |
| 5 | 6.5 | 10.83 | F | Idiopathic | ES | Bosentan, sildenafil, iloprost, nifedipine | Stent | 5 | 9 | 44 | 99 | 100 | 23 | 25 | No | No further syncope |
| 6 | 0.3 | 0.38 | F | Idiopathic | RRHF | Nitric oxide, sildenafil, epoprostenol | Stent | 10 | 8 | 62 | 94 | 97 | 10 | 25 | No | Initially class III then class II |
| 7 | 0.7 | 0.78 | M | Idiopathic | RRHF | Bosentan, sildenafil, iloprost | Stent | 4.5 | 16 | 79 | 98 | 85 | 11 | 13 | No | No improvement in functional classDied 35 days after AS |
| 8 | 1.8 | 1.89 | F | Idiopathic | RRHF | Bosentan, sildenafil | Stent | 3.5 | 12 | 56 | 100 | 76 | 11 | 9 | PH crisis | No improvement in functional classTransplantation at 13 months after AS |
| 9 | 2.7 | 4.68 | M | CHD | ES | Bosentan, sildenafil, iloprost | Stent | 5 | — | — | 100 | — | 2 | 24 | No | No exertional syncope (recurred at 4 years) |
| 10 | 0.2 | 2.78 | M | Familial | ES | Bosentan, sildenafil, iloprost, nifedipine | Stent | 10 | 11 | 87 | 99 | 91 | 9 | 15 | No | No further exertional syncopeTransplantation at 2 years and 11 months after AS |
| 11 | 0.08 | 1.68 | M | CHD | ES | Bosentan, sildenafil, iloprost | Surgical (IVC agenesis) | 6 | 10 | 43 | 98 | 98 | 9 | 10 | PH crisis | No further exertional syncopeTransplantation at 1 year and 3 months after AS |
AS, atrial septostomy; CHD, congenital heart disease; Dx, diagnosis; ES, exertional syncope; F, female; IVC, inferior vena cava; M, male; PAP, mean pulmonary arterial pressure; PH, pulmonary hypertension; RAP, mean right atrial pressure; RRHF, refractory right heart failure; TAPSE, tricuspid annular plane systolic excursion.
Five children (45.45%) had syncope, which resolved after AS, and 6 had signs of right ventricular hypertrophy despite optimized pulmonary vasodilator treatment (63.3% were on triple therapy). AS was performed as an interventional procedure in 10 (9 with a stent and 1 with angioplasty and balloon dilatation) and in 1 case, after a failed attempt in the catheterization laboratory, as a surgical procedure. The median size of the AS was 5 (3.5-12) mm.
There was no periprocedural mortality. Associated complications were severe PH crises during the procedure in 2 patients: 1 required adrenaline, inhaled nitric oxide, iloprost, and epoprostenol and then stabilized, and 1 required increased pulmonary vasodilator therapy. No significant changes were observed initially in the echocardiographic parameters of right ventricular remodeling or function, such as the sphericity index or interventricular septum type, but the tricuspid annular plane systolic excursion improved in 3 patients (27.2%).
The median follow-up was 25.85 months. The event-free survival (death and/or transplant) at 1 month was 100%; at 6 months, 72.72% (2 patients died due to disease progression without receiving a lung transplant); at 1 year, 72.72%; and at 2 years, 54.54% (4 patients received a transplant). The presence of a stent posed no added difficulty and it was removed during the same procedure.
AS in children with PH is a procedure that, despite the risk from the clinical condition, has low mortality and can help to improve symptoms with resolution of syncope, although it does not stop disease progression. It should be noted that, although this was a retrospective review, with a heterogeneous sample and a small number of patients, it is the largest series of children treated with this technique in Spain.
CONFLICTS OF INTERESTA. Moreno-Galdó has received fees for presentations from Abbvie and for attendance at conferences from Novartis and Actelion. D.C. Albert Brotons has received fees for conference attendance from Actelion.