Aortic stenosis (AS) and mitral regurgitation (MR) are the most prevalent valvular heart diseases in Western countries.1 The rate of concomitant significant MR in patients with severe AS ranges between 2% and 33%.2 Double-valve surgery has been associated with higher morbidity and mortality compared with isolated surgical aortic valve surgery, especially in elderly patients.3 However, this approach remains the standard of care for patients with combined mitral-aortic valve disease.
Transcatheter aortic valve replacement (TAVR) and transcatheter mitral valve repair, particularly edge-to-edge repair with the MitraClip device (Abbott Vascular, Inc. Santa Clara, CA, United States) have emerged as a treatment option for patients deemed inoperable or at high surgical risk. The combination of the 2 transcatheter therapies would appear to be a viable approach for managing high-risk patients with concomitant AS and MR. This systematic review seeks to analyze the safety and feasibility of combined transcatheter mitral-aortic valve treatment.
A comprehensive, systematic review was performed of published data in the English language describing double-valve (aortic and mitral) transcatheter therapy. A computerized search was conducted to identify all relevant studies from the PubMed, EMBASE, and Google Scholar databases using the following terms: “TAVR”, “TAVI”, “transcatheter/percutaneous aortic valve”, “transcatheter/percutaneous aortic valve replacement/implantation”, “transcatheter/percutaneous mitral valve repair”, “MitraClip”, and “transcatheter/percutaneous double-valve treatment/intervention”.
A total of 10 articles () describing 33 patients (mean age 79 ± 3 years, 69.7% male) were included. Baseline clinical characteristics and echocardiographic data are summarized in the Table. All patients were considered to be inoperable or at high risk for surgery. The indication for transcatheter aortic valve replacement was predominantly severe native AS, whereas 4 (12.1%) patients underwent TAVR to treat a degenerated surgical bioprosthesis. Moderate-to-severe MR was present before TAVR in all but 1 patient, who had an iatrogenic mitral chord rupture during TAVR, resulting in severe MR.
Baseline, Procedural Characteristics, In-hospital and Follow-up Outcomes
| N = 33 | |
|---|---|
| Baseline clinical characteristics | |
| Age | 79.4 ± 3.1 |
| Male | 23/33 (69.7) |
| NYHA class III-IV | 28/29 (96.6) |
| Logistic EuroSCORE, %a | 27.0 ± 13.0 |
| STS score, %b | 8.7 ± 6.9 |
| Prior CABG | 15/33 (45.5) |
| Prior surgical AVR | 4/33 (12.1) |
| Baseline echocardiography | |
| LVEF, % | 39.8 ± 7.0 |
| Mean aortic valve gradient, mmHg | 42.7 ± 19.5 |
| Aortic valve area, mm2 | 0.72 ± 0.15 |
| Functional mitral regurgitation | 13/30 (43.3) |
| Procedural characteristics | |
| TAVR before MitraClip | 29/33 (87.9) |
| Simultaneous TAVR and MitraClip | 3/33 (9.1) |
| TAVR after MitraClip | 1/33 (3.0) |
| Mean time between procedures, d | 172 ± 344 |
| TAVR | |
| Device success | 32/33 (97.0) |
| Balloon expandable valve | 14/32 (43.7) |
| Transfemoral approach | 22/31 (70.9) |
| Valve-in-valve | 4/33 (12.1) |
| Procedural complications | |
| Need for permanent pacemaker implantation | 8/33 (24.2) |
| Need for second valve implantation | 1/33 (3.0) |
| Mitral chord injury | 1/33 (3.0) |
| Acute kidney injury not requiring hemodialysis | 1/33 (3.0) |
| Transcatheter mitral valve repair (MitraClip) | |
| Technical successc | 28/30 (93.3) |
| Length of hospital stay, days | 5 ± 1 |
| ≥ 2+ MR post-MitraClip | 7/19 (36.9) |
| Procedural complications | |
| Cardiac tamponaded | 1/33 (3.0) |
| Clip embolization | 0/33 (0) |
| Minor bleeding from access sidee | 1/33 (3.0) |
| Transfusion (≥ 1 blood units) | 1/33 (3.0) |
| Length of hospital stay, d | 5 ± 1 |
| In-hospital outcomes | |
| Patients with any complication | 11/33 (33) |
| In-hospital MACE (death, MI, stroke) | 3/33 (9.1) |
| Deathf | 2/33 (6) |
| Major stroke | 1/33 (3) |
| Myocardial infarction | 0/33 (0) |
| Conversion to open heart surgery | 1/33 (3.0) |
| Follow-up | |
| Follow-up, d (range)g | 179.4 (14-390) |
| NYHA class at last follow-up ≥ III | 7/27 (25.9) |
| Rehospitalization | 5/33 (15.2) |
| 6-month mortalityh | 4/27 (14.8) |
AVR, aortic valve replacement; CABG, coronary artery bypass graft; LAA, left atrial appendage; LVEF, left ventricular ejection fraction; MACE, major adverse cardiovascular events; MI, myocardial infarction; MR, mitral regurgitation; NYHA, New York Heart Association; TAVR, transcatheter aortic valve replacement; STS, Society of Thoracic Surgeons.
The procedural details and in-hospital outcomes are described in Table. Transcatheter mitral valve repair was performed using the MitraClip system in all patients. Transcatheter aortic valve replacement was performed before the MitraClip procedure in 29 patients (87.9%); 27 of them were discharged after TAVR and had the MitraClip procedure at a mean of 172 ± 344 days post-TAVR. In 3 patients (9.0%), both valves were treated during the same intervention. Only 1 patient (3.0%) underwent MitraClip implantation before the TAVR procedure. Mitral regurgitation was significantly reduced in most patients, achieving MR grade ≤ 2+ in 28 out of 30 (93.3%) after the procedure.
Follow-up information (ranging from 14 days to 390 days) was available in the 27 patients; 3 patients (9.7%) out of the 31 who were discharged after MitraClip implantation died from a cardiac cause on days 17, 50, and 419 post-MitraClip repair. New York Heart Association (NYHA) functional class was reported for 27 patients; among these, 20 (60.6%) were in NYHA class I-II at the last follow-up. Five patients (15.2%) had to be rehospitalized due to heart failure. One patient underwent cardiac surgery with conventional aortic and mitral valve replacement as a consequence of aortic valve prosthesis migration and recurrent severe MR secondary to single mitral leaflet detachment. The main complications associated with both procedures are summarized in the Table.
This systematic review indicates that transcatheter double-valve treatment appears to be safe and feasible for high-surgical-risk patients with severe AS and concomitant severe MR. The in-hospital mortality appears to be slightly higher than that described in contemporary MitraClip registries.4,5 TAVR did not seem to increase the periprocedural risk or interfere with MitraClip implantation. One of the patients who died underwent TAVR and MitraClip implantation in a single session. The other case of in-hospital mortality was in a very high-risk patient (logistic EuroSCORE, 65%), who was in a critical clinical condition.
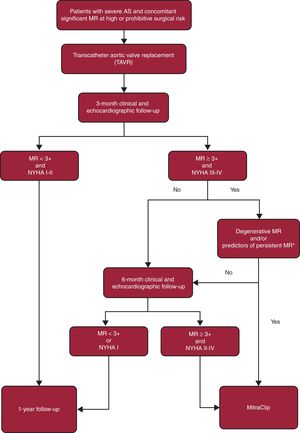
Recent studies have suggested that significant concomitant MR improves in approximately 50% to 60% of patients post-TAVR, especially MR of functional etiology.2 In this population at high-risk of periprocedural complications, it is important to decide the need for the 2 (aortic and mitral) procedures and their optimal timing, avoiding unnecessary risks in patients who could potentially show MR improvement during follow-up. Although there is no conclusive evidence in the literature regarding which strategy (staged vs single mitral and aortic procedure) is associated with superior outcomes, in our opinion, a staged approach addressing the aortic valve first would likely be the most appropriate strategy. After TAVR, a comprehensive clinical follow-up is essential for the early identification of patients who are unlikely to show spontaneous MR improvement in order to offer the possibility of transcatheter mitral repair. Based on the available data, we propose an algorithm (Figure) that could guide the clinical decision-making process in such patients.
Several limitations of the present review warrant consideration. First, inherent to all systematic reviews is the issue of publication bias, which could have limited the extent of clinical experience with combined transcatheter treatment for combined mitral and aortic valve disease available in the literature. Second, patients’ baseline characteristics, echocardiographic findings, and procedural-related data were reported heterogeneously among publications.
In conclusion, transcatheter double-valve treatment appears to be safe and feasible for high-surgical-risk patients with severe AS and concomitant severe MR. Larger studies are needed to confirm the safety and to establish the most appropriate timing for this double treatment strategy in this challenging group of patients who combine significant aortic and mitral valve disease.
.
A. Regueiro and T. Rodríguez-Gabella are supported by a grant from the Fundación Alfonso Martín Escudero (Madrid, Spain). J. Rodés-Cabau holds the Research Chair “Fondation Famille Jacques Larivière” for the Development of Structural Heart Disease Interventions.