Recent studies have shown that chronic inflammation associated with the development and aggravation of type 2 diabetes mellitus is an important factor in the pathogenesis of diabetic macrovascular complications. Diabetic cardiomyopathy is characterized by functional and structural cardiac changes, most frequently myocardial fibrosis, including myocardial cell death and accumulation of extracellular matrix protein.1 Mycophenolate mofetil (MMF) was shown to ameliorate renal injury and prevent the development of nephropathy through anti-inflammatory mechanisms and attenuated podocyte apoptosis.2 A study using a carotid artery balloon injury model revealed that MMF greatly reduced intimal hyperplasia with decreased expression of TGF-β1 and collagen III.3 Mycophenolate mofetil prevented endothelial dysfunction through enhanced nitric oxide availability with improvement of endothelial nitric oxide synthase.4 The purpose of this study was to evaluate its effect on diabetic cardiomyopathy resulting from type 2 diabetes mellitus.
Six-week-old male nondiabetic db/m and diabetic db/db mice were purchased from Jackson Laboratory (Sacramento, California, United States). All mice received a diet of rodent pellets (348 kcal/100 g) containing 5.5% crude fat and tap water ad libitum. Mycophenolate mofetil (Roche Pharma AG, Grenzach-Wyhlen, Germany) was incorporated into chow (Dooyeol Biotech, Seoul, Korea) at an oral dose of 30mg/kg body weight/d at a chow consumption rate of 0.2 g/g body weight. At 8 weeks of age, the mice were divided into 3 groups of 6 mice each as follows: the nondiabetic control group (db/m), the diabetic group (db/db), and the diabetes with MMF group (db/db + MMF). During experiments, food intake, water intake, urine volume, body weight, fasting plasma glucose level, and glycosylated hemoglobin were measured monthly. All animal experiments were performed in compliance with the guidelines of the Animal Research Ethics Committee of Kyung Hee University, Seoul, Korea.
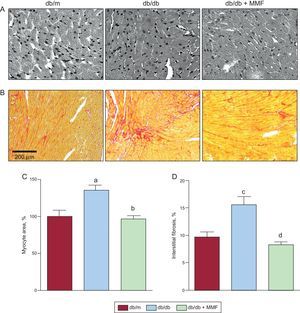
Transthoracic echocardiography was performed to obtain 2-dimensional, M-mode images using a 12-MHz linear probe prior to sacrificing the animal at the end of the study protocol (20 weeks) using standard formulae. The cardiac tissue was fixed in buffered 4% formaldehyde and embedded in paraffin. Tissue sections of 5 μm were deparaffinized, rehydrated and stained with hematoxylin and eosin to determine cardiomyocyte area. To visualize cardiac fibrosis, the sections were stained with Sirius red. Images from all stained cardiac tissue sections were analyzed using ImageJ software version 1.47t. Capillary and arteriolar densities in the myocardium were evaluated by means of CD31 and α-smooth muscle actin staining.
All values are expressed as means ± SE. Results were analyzed using the Kruskal-Wallis nonparametric test for multiple comparisons. Significant differences in the Kruskal-Wallis test were confirmed by the Mann-Whitney test (used to compare mean differences). P-values<.05 were considered statistically significant.
Various biochemical parameters such as body weight, daily food intake, and daily water intake were higher in db/db mice groups than in the db/m mice group, but there was no difference between the 2 db/db mice groups in these parameters. Both db/db mice groups showed higher glycosylated hemoglobin levels than the db/m mice group. In particular, glycosylated hemoglobin was higher in the MMF-treated db/db group at 20 weeks.
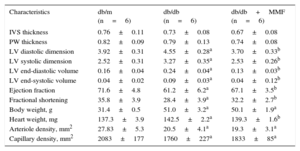
As shown in the Figure, the diabetic mouse heart developed hypertrophy, including increased cardiac myocyte area and interstitial fibrosis. Cardiomyocyte cross-sectional area was decreased in MMF-treated db/db mice compared with untreated db/db mice (Figures A-C). Furthermore, interstitial fibrosis was also attenuated in the MMF-treated db/db heart (Figures B-D). However, MMF had no effect on intramyocardial arteriole or capillary densities (Table). Echocardiographic studies show dilated left ventricle and reduced cardiac performance in db/db mice (Table). The MMF-treated group showed a significant reduction in left ventricle dilatation, as indicated by a lower left ventricular diastolic and systolic dimension and volume. The left ventricular ejection fraction and fractional shortening were improved by MMF.
Effects of MMF on diabetic cardiomyopathy in db/db mice. Mycophenolate mofetil attenuates hypertrophy and fibrosis of myocytes in db/db mice. Representative images of hematoxylin and eosin-stained cardiac tissues (A) and Sirius red-stained interstitial areas (B). Quantification of cardiomyocyte cross-sectional area (C) and percentage of interstitial (D) areas in the graph. Data were analyzed by 1-way ANOVA with Newman-Keuls post-hoc testing and expressed as mean ± SE (n ≥ 15 per group). MMF, mycophenolate mofetil.
aP < 0.001 vs db/m.
bP < 0.001 vs db/db.
cP < 0.01 vs db/m.
dP < 0.01 vs db/db.
Effects of Mycophenolate Mofetil on Diabetic Cardiomyopathy in db/db Mice
| Characteristics | db/m (n=6) | db/db (n=6) | db/db+MMF (n=6) |
|---|---|---|---|
| IVS thickness | 0.76±0.11 | 0.73±0.08 | 0.67±0.08 |
| PW thickness | 0.82±0.09 | 0.79±0.13 | 0.74±0.08 |
| LV diastolic dimension | 3.92±0.31 | 4.55±0.28a | 3.70±0.33b |
| LV systolic dimension | 2.52±0.31 | 3.27±0.35a | 2.53±0.26b |
| LV end-diastolic volume | 0.16±0.04 | 0.24±0.04a | 0.13±0.03b |
| LV end-systolic volume | 0.04±0.02 | 0.09±0.03a | 0.04±0.12b |
| Ejection fraction | 71.6±4.8 | 61.2±6.2a | 67.1±3.5b |
| Fractional shortening | 35.8±3.9 | 28.4±3.9a | 32.2±2.7b |
| Body weight, g | 31.4±0.5 | 51.0±3.2a | 50.1±1.9a |
| Heart weight, mg | 137.3±3.9 | 142.5±2.2a | 139.3±1.6b |
| Arteriole density, mm2 | 27.83±5.3 | 20.5±4.1a | 19.3±3.1a |
| Capillary density, mm2 | 2083±177 | 1760±227a | 1833±85a |
IVS, interventricular septum; LV, left ventricular; MMF, mycophenolate mofetil; PW, posterior wall.
Data are expressed as mean ± SE.
The results of this study suggest that MMF prevented the progression of diabetic cardiomyopathy in db/db mice independent of glycemic control. The MMF regimen is widely used in heart transplantation to prevent allograft vasculopathy, but limited data are available to demonstrate the beneficial effects of MMF on the nontransplanted heart. Some reports have suggested that MMF has antiatherogenic properties and that it downregulates endothelial adhesion molecules, alleviates proliferation of arterial smooth muscle cells, and induces apoptosis of monocytes or macrophages.2,5 A limitation of this study is that we did not evaluate the underlying cause of the cardioprotective effect of MMF. If confirmed in humans, the cardioprotective effect achieved by MMF in our small experience with a diabetic mouse model could be an effective way to alleviate cardiac hypertrophy and fibrosis in patients with diabetic cardiomyopathy.
FundingThis work was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MEST) (No. 2012M3A9C6050507). Mycophenolate mofetil was kindly donated by Roche.