A new laser balloon that allows visualization of atrial tissue has recently been introduced for pulmonary vein electrical isolation. The aim of this study was to evaluate the mid-term safety and efficacy of this catheter in the treatment of atrial fibrillation.
MethodsLaser balloon ablation was performed in 71 patients with paroxysmal (80%) or persistent (20%) atrial fibrillation. Arrhythmia recurrence was defined as any episode lasting longer than 30 seconds. During follow-up, regular visits were performed every 3 months with 24- to 48-hour Holter tests.
ResultsIsolation was possible in 275 of 278 (99%) of pulmonary veins. Mean procedure and fluoroscopy times were 154 ± 25 and 34 ± 15minutes, respectively. A total of 89% of veins were isolated during the first attempt. The most common complication was phrenic nerve paralysis (5.6%), which appeared in only the first 18 cases. A total of 59 patients received follow-up for a mean of 420 ± 193 days, with a rate of arrhythmia recurrence of 12% and 30%, respectively, in paroxysmal and persistent atrial fibrillation (P = .155).
ConclusionsThe laser balloon is a safe and effective system for pulmonary vein electrical isolation. Its advantages include the capacity to adapt to pulmonary vein anatomy using a single catheter, the efficacy with which pulmonary vein electrical isolation is achieved, and the favorable mid-term clinical progress, even for patients with persistent atrial fibrillation.
Keywords
Atrial fibrillation (AF) ablation is the recommended treatment for patients with paroxysmal or persistent AF who remain symptomatic despite antiarrhythmic therapy.1,2
In AF ablation, the main purpose is to achieve complete electrical isolation of pulmonary veins (PVs).3 This is usually accomplished by point-to-point radiofrequency ablation around the ostium of PVs using irrigated tip catheters and electroanatomical navigation systems. However, the procedure is complex and time-consuming and has a long learning curve, with outcomes highly dependent on the operator. These limitations have led to the development of catheters specially designed to achieve PV electrical isolation using other energy sources.4,5 Balloon cryoablation is the alternative to radiofrequency used at most hospitals and for which there is more clinical experience and scientific evidence. At present, point-to-point radiofrequency ablation and cryoablation are the 2 most common methods used for AF ablation.3
In recent years, a new ablation system that uses laser energy as an ablation source has been added to the therapeutic armamentarium.6–9 The catheter is fitted with an adaptable balloon and is the first system to allow direct visualization of the left atrium and PVs to guide the ablation. Early clinical results show a good safety and efficacy profile, as well as a greater durability of PV isolation.8,9
The aim of this study was to describe the experience at our facility, to evaluate the efficacy and safety of this laser catheter in a series of patients referred to our hospital for ablation of paroxysmal or persistent AF, and to report on clinical outcomes over a mean follow-up of 1 year.
METHODSPatientsThe laser balloon ablation system first became available for use in our hospital in February 2013. As of that date, patients referred for AF ablation were alternatively assigned to treatment by radiofrequency ablation or laser ablation if they met either of the following criteria: a) patients with symptomatic paroxysmal AF who were refractory to at least 1 antiarrhythmic drug, and b) as of September 2013, patients with persistent AF from < 1 year previously who were symptomatic and refractory to at least 1 antiarrhythmic drug.
Patients with any of the following characteristics were excluded: a) left ventricular ejection fraction < 50%; b) age < 18 years or > 75 years; c) concomitant significant structural heart disease, and d) left atrial diameter > 50 mm. The study obtained results from 71 patients with AF who had been treated by laser-catheter ablation, from among a total of 137 patients referred for AF ablation.
Description of the Laser BalloonThe ablation system (CardioFocus, Inc.; Marlborough, Maryland, United States) includes a deflectable sheath (inner and outer diameters, 12 Fr and 15 Fr), balloon catheter with an inflatable diameter, endoscope, and console.
At its distal end, the balloon has a soft, nontraumatic tip to ease insertion of the balloon catheter into each PV and to reduce the risk of traumatic injury. The balloon diameter is adaptable and can progressively inflate up to a maximum of 38 mm, in order to maximize PV-to-balloon contact. The catheter shaft is multi-lumen for D2O circulation to cool the balloon, for real-time visualization of the outer balloon surface via a 2-Fr endoscope, and for an optical fiber used to generate a movable light beam that covers 30°. Laser energy (980-nm diode laser) is administered through the same optical fiber. Endoscopic vision is partially obstructed in the area behind the central shaft of the balloon. Once ablation is completed in the visible tissue around the PV, the balloon is ruptured to complete the ablation circumference.
The console controls several parameters, among them, the energy administered (5.5-12 W), application time (20 or 30 s), and balloon diameter. The console has 2 images: a real-time image and a side-by-side image of previous and current applications to ensure continuity of the ablation line.
Ablation ProcedureTwo operators (J. Osca and O. Cano) handled all cases simultaneously. All cases were treated by cannulation of the coronary sinus with a decapolar catheter (reference for transseptal puncture and right diaphragmatic stimulation) and use of an esophageal thermometer (SensiThermTM, St. Jude Medical; Minnesota, United States). Double transseptal puncture was performed by a modified Brockenbrough technique using 2 sheaths of 8.5 Fr (SL1; St. Jude Medical). A decapolar circular catheter was inserted through 1 of the sheaths to map the PVs (LassoTM, Biosense Webster; Diamond Bar, California, United States), whereas the free SL1 sheath was switched to a deflectable sheath (CardioFocus), always infused with heparinized solution. After transseptal puncture and throughout the rest of the procedure, heparin was administered to achieve an activated coagulation time > 300 s.
Selective angiography was then performed on the PVs, and after venography, the laser balloon was introduced through the deflectable sheath and advanced to the PV to start ablation. The purpose of ablation was to focus the laser beam on the area closest to the antrum, to the extent possible. Each application was overlapped with the preceding application by 30% to 50% to achieve a continuous ablation line. The energy administered was adjusted according to the anterior or posterior position of the application (8.5 W in the posterior wall; 10–12 W in the anterior wall), according to the width of the tissue visualized (higher power for wider tissues), and by reducing the power in the case of blood retained or trapped by the balloon. Once ablation was completed around each PV, the presence of bidirectional conduction block was confirmed.
Due to persistent conduction between the PV and the left atrium, additional laser applications were performed according to the records obtained with the LassoTM catheter.
Once the persistence of PV electrical isolation in the next 20minutes was confirmed, the procedure was finished. Adenosine was not used in any patients to confirm reconnection.
Postablation Follow-upAfter ablation, patients received follow-up in the arrhythmia outpatient clinic every 3 months. The information obtained during follow-up included an electrocardiogram and a 24- to 48-hour Holter at each visit. Recurrence was not considered to include arrhythmias that appeared within the first 3 months after ablation (blind period). Antiarrhythmic therapy was discontinued thereafter and anticoagulant therapy was maintained according to CHA2DS2-VASc score.
The primary efficacy endpoint of ablation was AF/atrial tachycardia recurrence-free survival during follow-up. Recurrence was defined as arrhythmia episodes > 30 s on Holter monitoring or conventional electrocardiogram.
Statistical AnalysisThe statistical analysis was performed with SPSS 15.0. Categorical variables are expressed as a percentage. Continuous variables are expressed as mean ± standard deviation.
The categorical variables were compared using the chi-squared test. Quantitative variables were compared using the Student t test and ANOVA.
Kaplan-Meier curves were used to analyze AF/atrial tachycardia recurrence-free survival, and the log rank test was used to compare survival curves for patients with paroxysmal or persistent AF. P values < 0.05 were considered statistically significant.
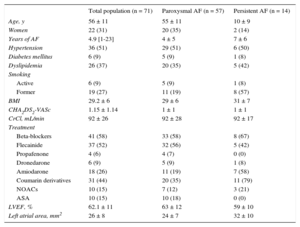
ResultsPatient CharacteristicsLaser balloon ablation was performed in 71 patients diagnosed with paroxysmal (57 patients) or persistent (14 patients) AF and referred between February 2013 and March 2015 for ablation based on clinical criteria. Mean age was 56 ± 11 years (women, 31%), and all patients had been unsuccessfully treated with at least 1 antiarrhythmic agent. The other characteristics of the sample are listed in Table 1. A total of 6% had a history of typical atrial flutter (prior ablation of cavotricuspid isthmus) and 1 (1.4%) patient had undergone AF radiofrequency ablation.
Population Characteristics
| Total population (n = 71) | Paroxysmal AF (n = 57) | Persistent AF (n = 14) | |
|---|---|---|---|
| Age, y | 56 ± 11 | 55 ± 11 | 10 ± 9 |
| Women | 22 (31) | 20 (35) | 2 (14) |
| Years of AF | 4.9 [1-23] | 4 ± 5 | 7 ± 6 |
| Hypertension | 36 (51) | 29 (51) | 6 (50) |
| Diabetes mellitus | 6 (9) | 5 (9) | 1 (8) |
| Dyslipidemia | 26 (37) | 20 (35) | 5 (42) |
| Smoking | |||
| Active | 6 (9) | 5 (9) | 1 (8) |
| Former | 19 (27) | 11 (19) | 8 (57) |
| BMI | 29.2 ± 6 | 29 ± 6 | 31 ± 7 |
| CHA2DS2-VASc | 1.15 ± 1.14 | 1 ± 1 | 1 ± 1 |
| CrCl, mL/min | 92 ± 26 | 92 ± 28 | 92 ± 17 |
| Treatment | |||
| Beta-blockers | 41 (58) | 33 (58) | 8 (67) |
| Flecainide | 37 (52) | 32 (56) | 5 (42) |
| Propafenone | 4 (6) | 4 (7) | 0 (0) |
| Dronedarone | 6 (9) | 5 (9) | 1 (8) |
| Amiodarone | 18 (26) | 11 (19) | 7 (58) |
| Coumarin derivatives | 31 (44) | 20 (35) | 11 (79) |
| NOACs | 10 (15) | 7 (12) | 3 (21) |
| ASA | 10 (15) | 10 (18) | 0 (0) |
| LVEF, % | 62.1 ± 11 | 63 ± 12 | 59 ± 10 |
| Left atrial area, mm2 | 26 ± 8 | 24 ± 7 | 32 ± 10 |
ASA, acetylsalicylic acid; AF, atrial fibrillation; BMI, body mass index; CHA2DS2-VASc, congestive heart failure, hypertension, age ≥ 75 (doubled), diabetes, stroke (doubled)-vascular disease and sex category (female); CrCl, creatinine clearance; LVEF, left ventricular ejection fraction; NOACs, new oral anticoagulants.
Data are expressed as No. (%) or mean ± standard deviation or median [range].
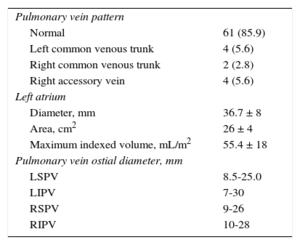
The PV pattern was assessed by magnetic resonance imaging in 78% of cases and by angiography in all patients during the actual ablation procedure (Table 2). Imaging revealed a conventional position in 85.9% of patients, a left common trunk in 5.6%, a right common trunk in 2.8%, and another pattern (right accessory vein) in 5.6% of cases.
Vein Anatomy
| Pulmonary vein pattern | |
| Normal | 61 (85.9) |
| Left common venous trunk | 4 (5.6) |
| Right common venous trunk | 2 (2.8) |
| Right accessory vein | 4 (5.6) |
| Left atrium | |
| Diameter, mm | 36.7 ± 8 |
| Area, cm2 | 26 ± 4 |
| Maximum indexed volume, mL/m2 | 55.4 ± 18 |
| Pulmonary vein ostial diameter, mm | |
| LSPV | 8.5-25.0 |
| LIPV | 7-30 |
| RSPV | 9-26 |
| RIPV | 10-28 |
LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior RIPV, right superior pulmonary vein.
Data are expressed as No. (%) or mean ± standard deviation or ranges.
The procedure data are listed in Tables 3 and 4. The mean procedure duration (from catheter introduction to withdrawal) was 154 ± 25minutes, with a mean fluoroscopy time of 34.4 ± 14minutes. Isolation was possible in 275 of 278 (98.9%) PVs, and isolation of all veins was achieved in 96% of patients. The isolated veins included 100% of all veins, except the right inferior PVs, in which case 95.6% (65 of 68) were isolated. Effective isolation was achieved in all cases of common venous trunk, whether right or left. In 89.2% of PVs, electrical isolation was achieved with completion of the first line of ablation. In the remaining 10.8%, additional applications were necessary. The mean time used for isolation of each vein, considered to be the time from balloon introduction to vein isolation, was 16 to 19 minutes per vein. The left common trunk was the venous structure requiring the highest number of applications and the longest ablation times to achieve isolation. The mean power level used and the mean number of lesions needed to isolate each vein are listed in Table 4.
Ablation Data (n = 70)
| Veins isolated | 275/278 (98.9) |
| Veins isolated at first attempt | 248/278 (89.2) |
| Attempts per vein | |
| RSPV | 1.12 ± 0.4 |
| RIPV | 1.13 ± 0.4 |
| Right common trunk | 1 ± 0 |
| LSPV | 1.2 ± 0.5 |
| LIPV | 1 ± 0 |
| Left common trunk | 1.5 ± 1 |
| Ablation lesions per patient | 113.15 ± 28 |
| Fluoroscopy time, min | 34.4 ± 14.7 [9-81] |
| Procedure time, min | 154 ± 25 [94-220] |
| Time to insulation, min | |
| RSPV | 18.3 ± 10.7 |
| RIPV | 17.9 ± 10 |
| Right common trunk | 16.5 ± 0.7 |
| LSPV | 18.8 ± 7.2 |
| LIPV | 15.7 ± 6.1 |
| Left common trunk | 43.3 ± 16.4 |
LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
Data are expressed as no./No. (%) or mean ± standard deviation [range].
Number and Type of Pulmonary Veins Isolated (n = 278)
| PVs | PVs isolated by laser | PVs isolated at first attempt | Applied power, W | Applications per vein | |
|---|---|---|---|---|---|
| RSPV | 68 | 68 (100) | 61 (89.7) | 9.6 ± 0.9 | 30 ± 11 |
| RIPV | 68 | 65 (95.6) | 57 (83.8) | 9.1 ± 1 | 26 ± 10 |
| RCT | 2 | 2 (100) | 2 (100) | 9 ± 1.41 | 31 ± 10 |
| LSPV | 66 | 66 (100) | 55 (83.3) | 9.5 ± 0.7 | 33 ± 11 |
| LIPV | 66 | 66 (100) | 66 (100) | 9.4 ± 0.8 | 28 ± 8 |
| LCT | 4 | 4 (100) | 3 (75) | 9.25 ± 0.4 | 71 ± 30 |
| Other (right accessory vein) | 4 | 4 (100) | 4 (100) | 8.6 ± 0.6 | 20 ± 7 |
LCT, left common trunk; LIPV, left inferior pulmonary vein; LSPV, left superior pulmonary vein; PVs, pulmonary veins; RCT, right common trunk; RIPV, right inferior pulmonary vein; RSPV, right superior pulmonary vein.
Data are expressed as No. (%) or mean ± standard deviation.
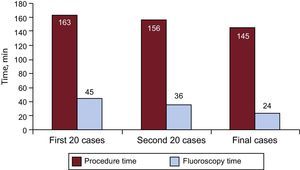
The learning curve was assessed by analyzing the procedure time every 20 patients (Figure 1). The total procedure duration and fluoroscopy time were shortened by an average of 10 minutes every 20 cases, differences that were statistically significant in the case of fluoroscopy time (P < .0001). The reduction in times plateaued at patient #40, and remained stable at procedure and fluoroscopy times of around 145 and 25 minutes, respectively.
ComplicationsThree (4.2%) patients had minor femoral bruising that resolved with noninvasive treatment, and 1 (1.4%) patient had a femoral pseudoaneurysm that required surgery. Two patients (2.8%, cases #23 and #54) had pericardial effusion related to manipulation of the ablation sheath: in one, this occurred during ablation attempts on the right inferior PV (after complete ablation of the other veins) and resolved with pericardiocentesis, and in the other, the procedure was complicated after sheath introduction in the left atrium and before ablation began. The latter required surgery for laceration of the base of the left atrial appendage.
Four (5.6%) patients experienced right phrenic nerve paralysis at the end of procedure. These cases appeared in the first 18 ablations undertaken, but there were no additional cases of diaphragmatic paralysis thereafter. Due to the complication, a change was made in the ablation techniques for the right superior PV, with particular attention given to performing antral ablation and controlling the power at a maximum of 10 W in the anterosuperior segment of the right superior PV.
There were no cases of atrial-esophageal fistula; 6 (8.5%) patients showed elevated esophageal temperature during laser application to the posterior wall. This finding required a change to the ablation line (toward the interior or exterior of the vein) and a reduction in the energy applied but did not hinder PV isolation in any case. No patient experienced stroke.
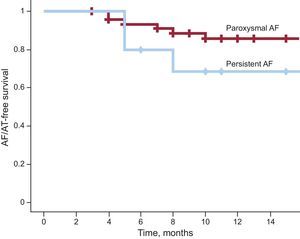
Clinical Result During Follow-upThe first 59 patients treated were followed for a mean period of 420 ± 193 (range, 121-753) days; at the time of follow-up, 30% were receiving antiarrhythmic therapy. A total of 12% of patients with paroxysmal AF had at least 1 recurrence of AF/atrial tachycardia, compared with 30% of patients with persistent AF (P = .155). Figure 2 shows the AF/atrial tachycardia recurrence-free survival curves (Kaplan-Meier) for both patient groups. Patients with paroxysmal AF showed greater arrhythmia recurrence-free survival: 21 (95% confidence interval, 19.6-23.4) months, compared with 12.9 (95% confidence interval, 9.9-15.8) months (log rank test, P = .20).
One patient was treated by a new ablation procedure 4 months after the initial ablation, due to recurrent atrial flutter crises (not observed before the initial ablation). During the study, isthmus-dependent flutter was induced and then ablated. Lastly, the left atrium was accessed to confirm electrical reconnection of the left inferior PV only, followed by point-by-point isolation of the floor of the vein antrum. One of the other patients was pending repeated ablation, whereas all others had arrhythmia recurrence while not receiving antiarrhythmics but became stable and asymptomatic once they were reintroduced. In keeping with patients’ preferences, a new ablation procedure was not ordered.
During follow-up, no additional complications were detected other than those at 24 hours after ablation, and recovery from diaphragmatic paralysis was observed in 3 of the 4 cases.
DISCUSSIONThis study describes the experience at our hospital with a balloon catheter designed for PV electrical isolation by laser in a series of 71 patients with paroxysmal or persistent AF. The main findings of the study were as follows: a) the laser balloon was highly effective in achieving PV electrical isolation; b) its design allowed adaptation to different PV anatomies with a single catheter; c) the device had a rapid learning curve; d) the main complication associated with use was phrenic nerve paralysis, with overall complication rates similar to those published with other ablation systems, and e) good mid-term clinical efficacy is achieved, with arrhythmia recurrence-free survival > 85% in paroxysmal AF and > 70% in persistent AF.
Short-term EfficacyThe purpose of paroxysmal AF ablation is PV electrical isolation. Several recently published studies provide definitive evidence on the need to achieve complete bidirectional conduction block to reduce AF recurrence. Similarly, the COR10 and GAP-AF11 studies compared an ablation procedure to achieve complete PV isolation compared with a simplified ablation procedure aiming at incomplete isolation. In both studies, the most important independent predictor of arrhythmia recurrence was the absence of complete isolation of all PVs during the initial ablation procedure.
Ablation in persistent AF is less standardized, however, and the guidelines recommend substrate ablation additional to pulmonary electrical isolation.3 In this context, the role of the balloon catheter would be less defined. Nonetheless, there is no clear evidence of the superiority of more extensive ablation in persistent AF. In addition, the STAR AF II study12 raises questions regarding the performance of ablation lines or the elimination of fragmented potentials and confirms that the main purpose of ablation of persistent AF, as in the case of paroxysmal AF, is to achieve permanent PV electrical isolation. Consequently, the balloon catheter could also play a therapeutic role in persistent AF.
The laser ablation catheter is an alternative to conventional ablation using radiofrequency or cryoablation. In our experience, which also considered the learning curve with this system, the balloon catheter adapted to each patient's vein anatomy allowed successful ablation of most veins and all venous trunks. These outcomes reflect the capacity of the laser balloon to modify its diameter by adapting itself to the diameter of each PV, including PV trunks and smaller-diameter veins. These findings are consistent with those published by other groups. In addition, a multicenter registry that collected experience with the first 200 patients treated by laser balloon achieved a short-term ablation success rate of 98.8% of PVs.9 All these results show that the laser balloon is an effective system to achieve complete PV isolation.
Procedure TimesIn our series, the mean ablation procedure and fluoroscopy times were 155 ± 225minutes and 34 ± 14 minutes, respectively, similar values to those previously described for AF ablation by laser catheter.7–9 Likewise, the procedure duration was similar or even shorter than that published for other AF ablation techniques.13,14
Lastly, a reduction in the mean procedure and fluoroscopy times of around 10 minutes was observed every 20 procedures performed, with stable times achieved around 145 and 25 minutes, respectively.
SafetyIn our series, the most common complication was diaphragmatic paralysis, which affected 4 (5%) patients. However, the disorder was transient, and recovery was observed in 3 of the 4 cases. This figure is slightly higher than that published by other studies that have evaluated the laser balloon (incidence, 2%-4.5%). In our study, the learning curve for the technique was seen to be related, as all diaphragmatic paralyses appeared in the first 18 cases.
Diaphragmatic paralysis is a known complication of other cardiac ablation procedures.14,15 For cryoablation, the main complication is AF, with published figures of up to 10%.14 This complication has been related to the use of the smallest cryoablation balloon located in the right superior PV, which would be associated with a higher risk of cooling of the adjacent anatomic structures. Likewise, excessive balloon pressure against the atrium at the juncture with the right superior PV could shorten the distance to the phrenic nerve.16 Both hypotheses may also explain the appearance of diaphragmatic paralysis during laser balloon ablation. Once we changed our practice in ablation of the right superior PV, there were no new episodes of phrenic nerve paralysis.
The second most common complication was the appearance of vascular complications, most without adverse consequences. Only 1 (1.4%) patient required surgery for a femoral pseudoaneurysm, with good postoperative outcome.
The most important complication was pericardial effusion in 1 patient who required surgical treatment.
There were no cases of stroke or transient ischemic attack. Other studies with the laser balloon have shown a low incidence of stroke and peripheral embolism. A multicenter series with 200 patients reported no strokes, and a study to compare cryoablation with laser-balloon ablation observed only 1 transient ischemic attack with the latter technique.9,16 Apart from diaphragmatic paralysis, these figures are consistent with those published previously, with an incidence of relevant complications after the ablation procedure around 4% to 4.5%.17
Mid-term Clinical ResultsIn our series, laser-catheter ablation was associated with an arrhythmia recurrence-free survival during a mean follow-up > 1 year of 88% of patients with paroxysmal AF and 70% of patients with persistent AF. Compared with the results from early studies on the laser balloon (around 65% of recurrence-free survival at 12 months of follow-up),8,9 our study observed greater mid-term efficacy. These positive results may be due to the routine use of more energy in areas at higher risk of electric reconnection, particularly at the ridge between the left atrial appendage and the left PV (applying 10 to 12 W). In addition, previous studies with the laser balloon have identified excessively ostial ablation as a possible cause of arrhythmia recurrence. In our series, particularly after the first few cases, particular attention was paid to applying the laser in a proximal position in the vein antrum. It was also impossible to exclude differences in the populations assessed due to the differences found. However, the overall results of our series were obtained in a population that also included patients with persistent AF who had been excluded in the initial studies.
Lastly, we have no data to indicate the superiority of any particular ablation method. The results obtained with the laser catheter in our series were at least as good as published results obtained at highly experienced facilities using radiofrequency or second-generation cryoballoon. Only 1 study has compared laser ablation with cryoablation in patients with paroxysmal AF.14 In that study, both strategies showed similar results after a 1-year follow-up. However, a tendency toward greater recurrent-free survival was observed in the laser treatment group, which had a lower incidence of reconnected veins after the performance of a second ablation procedure.
LimitationsThis study reflects early experience obtained with the laser ablation catheter at a single facility, with no comparison to a control group. Prolonged monitoring (7-day Holter or insertable Holter) was not performed and, therefore, arrhythmia recurrences could have been underestimated, particularly due to asymptomatic episodes. Moreover, the small sample size requires that larger prospective and randomized studies with various ablation techniques be conducted to evaluate possible differences in efficacy and to determine which subgroups might benefit from particular techniques.
CONCLUSIONSThe laser balloon is a safe and effective system for PV electrical isolation. Its advantages include the capacity to adapt to PV anatomy using a single catheter, the efficacy of pulmonary electrical isolation, the good mid-term outcomes in patients with paroxysmal AF, the promising results in patients with persistent AF (which should be confirmed in future studies), and the overall complication rates, which are similar to those published with other ablation systems.
CONFLICTS OF INTERESTNone declared.