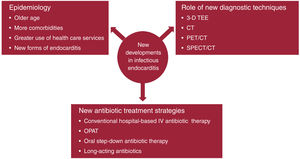
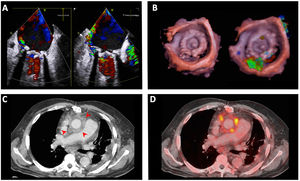
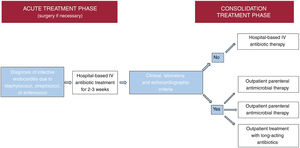
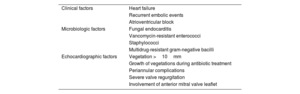
Infective endocarditis is a continually evolving disease. Present-day patients differ significantly from those treated a few decades ago: they tend to be older and have more comorbidities and health care-related episodes, while new groups of patients have emerged with new types of endocarditis, such as those affecting patients with percutaneous valve prostheses. There have also been changes in diagnostic techniques. Although transthoracic and transesophageal echocardiography are still the most commonly used imaging modalities, other techniques, such as 3-dimensional transesophageal ultrasound, cardiac computed tomography, and nuclear medicine tests (PET/CT and SPECT/CT), are increasingly used for diagnosing both the disease and its complications. In recent years, there have also been significant developments in antibiotic therapy. Currently, several treatment strategies are available to shorten the hospital phase of the disease in selected patients, which can reduce the complications associated with hospitalization, improve the quality of life of patients and their families, and reduce the health care costs of the disease. This review discusses the main recent epidemiological, diagnostic and therapeutic developments in infective endocarditis.
Keywords
Identify yourself
Not yet a subscriber to the journal?
Purchase access to the article
By purchasing the article, the PDF of the same can be downloaded
Price: 19,34 €
Phone for incidents
Monday to Friday from 9am to 6pm (GMT+1) except for the months of July and August, which will be from 9am to 3pm