Cryoballoon ablation of the pulmonary veins is a new technique that has proven useful in preventing paroxysmal and persistent atrial fibrillation recurrence. One of the most serious complications of this method is right phrenic nerve palsy. The usefulness of multidetector computed tomography to locate the right phrenic nerve and artery and predict the risk of phrenic nerve palsy during cryoablation according to the distance between the right phrenic neurovascular bundle and the right superior pulmonary vein ostium has recently been described. Fifty-five consecutive patients with paroxysmal atrial fibrillation (52±12 years) underwent balloon cryoablation, following multidetector computed tomography to measure the pulmonary veins. We were able to identify segments of the right pericardiacophrenic artery (mean length 25mm [range 7-68mm]) in only 10 patients (20%).
Keywords
Cryoballoon ablation of the pulmonary veins (PV) is an increasingly used approach that appears to yield similar outcomes to those of radiofrequency ablation1, 2, 3 for the prevention of recurrences of paroxysmal and persistent atrial fibrillation (AF), and with a lower incidence of complications. One of the most serious procedural complications is right phrenic nerve (RPN) palsy. A recent study has described the usefulness of multidetector computed tomography (MDCT) to consistently locate the course of the right phrenic artery and RPN, and to predict the risk of subsequent phrenic nerve palsy during cryoablation according to the distance between the right pericardiacophrenic artery (RPPA) and the right superior pulmonary vein (RSPV) ostium.4 The aim of the present study was to assess the usefulness of MDCT in locating the RPN in patients referred to PV ablation.
METHODSWe retrospectively analyzed consecutive patients referred to cryoballoon ablation of the PV and previous MDCT to measure PV diameter. A 64-slice computed tomography scanner was used in all studies (General Electric Medical Systems, Milwaukee, United States). Patients with a heart rate >60 bpm were administered 2 mg-10mg atenolol intravenously 5min before image acquisition. Electrocardiography-synchronized MDCT angiography was performed after the administration of non-ionic iodinated contrast media (Ultravist 370) which was adjusted according to weight.
Acquisition ParametersDetector collimation was 64×0.625mm, 120kV, 350-70mA with dose modulation and maximum intensity projection during 40%-80% of the cardiac cycle; gantry rotation time, 0.4s; pitch, 0.2-0.4 adapted to heart rate; and field of view, 140-180mm. Images were acquired during inspiration. The data were reconstructed at the mid-diastolic phase of the cardiac cycle using a retrospective ECG synchronization algorithm. The estimated dose of radiation was 12-20 mSv. The data were transferred to a workstation (General Electric Medical Systems) for assessment and postprocessing. A cardiologist and a radiologist, both experts in cardiac imaging (more than 120 cardiac MDCT studies per year) analyzed the course of the RPPA in axial slices, maximum intensity projection reconstructions and three-dimensional volume rendering, identifying it at the RSPV ostium. The outcomes were expressed in terms of visible or non-visible arteries. The variables are expressed as percentages or mean±standard deviation. Inter-rater agreement on the identification of the RPPA was measured using the kappa statistic. In case of inter-rater disagreement, identification was by consensus. The Student t-test was used to compare means. All statistical analyses were performed using the SPSS 15.0 software package.
RESULTSA total of 55 patients were included: 39 men (71%; mean age 52±12 years) with a structurally normal heart and paroxysmal AF. The RPPA was identified in 10 cases (20%) (observer 1, 14 cases; observer 2, 10 cases; concordance, κ=0.57). The complete artery could only be identified in 1 case (Figure 1, Figure 2) and only segments were identified in the remaining cases (mean size, 25mm [7-68mm]) (Figure 3); however, their relationship to the RSPV ostium (mean distance,18.9mm [3-26mm]) was identified in all cases. Three cases of right phrenic nerve palsy occurred during the procedure; in 2 of these it was possible to visualize the RPPA and measure its distance to the RSPV ostium (3mm and 13mm, respectively). The patients in whom the RPPA could be visualized tended to have a greater body mass index (BMI) (29.29±3.35 in those with visible arteries and 27.53±2.57 in those with non-visible arteries; P=.06).
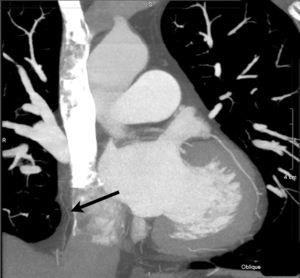
Figure 1. Coronal reconstruction using maximum intensity projection showing the course of the right pericardiacophrenic artery through the pericardial fat.
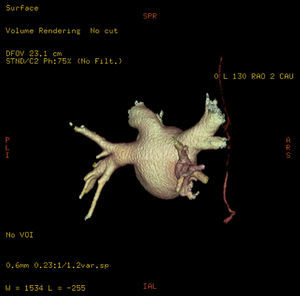
Figure 2. Three-dimensional reconstruction of left atrial volume and pulmonary veins, visualizing the right pericardiacophrenic artery (right posterior oblique view).
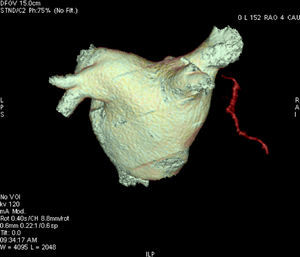
Figure 3. Three-dimensional reconstruction of left atrial volume and pulmonary veins, visualizing a fragment of the right pericardiacophrenic artery.
DISCUSSIONThe most serious complication of PV ablation is probably total or partial diaphragmatic paralysis due to phrenic nerve injury. This is more common during a cryoballoon isolation procedure (4%-10%)5, 6 than during radiofrequency ablation (0.10%-0.48%).7 Although it is usually reversible (80% per year),7, 8 very symptomatic patients have been described.9 The RPN runs close to the RSPV and the superior vein cava.10 Their ablation carries a high risk of injuring the RPN,8 although this has also been described during ablation of the right inferior PV.2 The conventional approach to prevent injury to the RPN is to pace it from the superior vein cava when delivering energy to the right PV and discontinue delivery early after detecting a decrease in or absence of diaphragmatic contraction.2, 6 A technique using MDCT angiography for identifying patients at high risk of phrenic nerve palsy has recently been described.4 The pericardiacophrenic artery and vein and the phrenic nerve run in parallel in a common neurovascular bundle that serves to locate all the structures by identifying any of them. Although the nerve has the same radiological density as the adjacent structures, by using MDCT and contrast enhancement of the RPPA, Matsumoto et al.11 observed this artery in 47% of patients. Horton et al.4 used this technique to identify the neurovascular bundle that contains the RPN and to measure its distance to the RSPV ostium, and they observed that when this was closer there was a significantly greater risk of phrenic palsy. Thus, this technique could be used to identify patients whose anatomy is more vulnerable to this type of complication during cryoablation procedures. Horton et al.4 identified the RPPA by applying the conventional protocols used to study the coronary arteries. We used the same protocols in our series, and although we were able to identify the course of the RPPA in a small percentage of patients, the complete artery could only be identified in 1 case. Thus, sensitivity was lower than that reported by Matsumoto et al.11 (47%) and much lower than the 100% reported by Horton et al.4 in their series. There is no obvious explanation for these differences, although the fact that the size of the arteries, less than 1 mm, was at the limit of the spatial resolution of the MDCT system could be responsible. Our results indicate that greater BMI could be associated with better visualization, but other authors have not observed this possible association.11 The potential use of nitrogylcerin to increase vessel diameter before image acquisition, which forms part of the protocol in the study of coronary disease, has not been described in the published series.4, 11 In any case, inter-rater agreement in the identification of the RPPA appears to be low, which demonstrates the difficulty in identifying this structure. In conclusion, given the technology and method used in this study, the sensitivity of the technique for the detection of the RPPA is low and is thus of limited use in the prediction of phrenic nerve injury during cryoablation. Thus, MDCT in itself is not justified as the imaging technique of choice in these patients, particularly when the dose of radiation they are exposed to and the option of using magnetic resonance imaging as an alternative are taken into account.
CONFLICTS OF INTERESTNone declared.
Received 21 November 2010
Accepted 10 February 2011
Corresponding author: Ctra. Madrid-Cartagena s/n, 30120 El Palmar, Murcia, Spain. jnieto1980@hotmail.com