In recent decades, animal experiments have shown numerous cardiovascular benefits for the hormone relaxin, traditionally known for its effects during pregnancy1. Relaxin lowers peripheral vascular resistance, reduces pulmonary congestion, improves cardiac output, and increases renal flow. By increasing nitric oxide, it produces vasodilating, anti-inflammatory, antiplatelet, and antioxidant effects. In addition, there have been descriptions of relaxin cardiac receptors and release in atria and ventricles2. Serelaxin, a recombinant form of endogenous relaxin, has emerged as a therapeutic option in acute heart failure (AHF) and shows promising results in the RELAX-AHF trial3. However, the role of relaxin in the pathophysiology of AHF has not been established, and there are only a few published clinical studies4–6. To date, it has not been shown that endogenous relaxin production is an important compensatory mechanism, a useful biomarker, or a hormone with prognostic value in patients with AHF.
The aim of our study was to analyze the role of relaxin as a neurohormonal mediator in patients with AHF. To conduct the research, we selected 43 consecutive patients (age, 69.8±9.4 years; men, 63%) who had been hospitalized with a diagnosis of AHF. Plasma relaxin concentrations were determined upon arrival at the emergency room and at 30 days after discharge and in stable condition. All concentrations were measured using a previously validated enzyme-linked immunoassay (Immunodiagnostik; Bensheim, Germany)2,4–6. The patients’ clinical, analytical, and echocardiographic variables were retrospectively collected from their medical history.
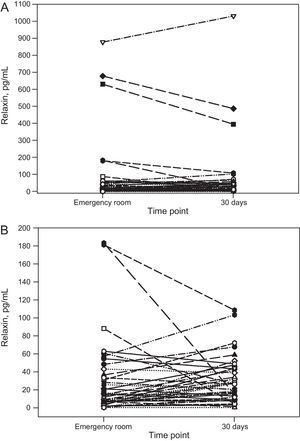
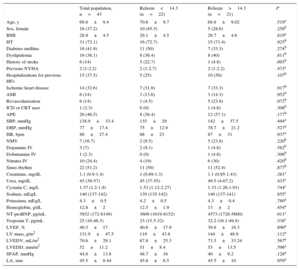
On arrival at the emergency room, relaxin was undetectable in 3 patients (7%); the median for all other patients was 14.3 [interquartile range, 5.8-48.0] (range, 1.5-878) pg/mL. Table lists the characteristics for the entire study population and according to median concentration. No significant associations were found with any clinical variables, including age, sex, left ventricular ejection fraction, N-terminal pro-brain natriuretic peptide, echocardiographic variables, and New York Heart Association functional class. After the acute event, relaxin concentration at 30 days had a median of 26.5 [11.4-47.3] (0.85-1031) pg/mL and strongly correlated with concentration on admission (rs=0.536; P<.001). The repeated measures analysis showed no significant change between measurements in the emergency room and at 30 days (P=.204, Wilcoxon test) or a uniform pattern of kinetics (Figure). During follow-up (median, 654 [332-932] days), 8 of 21 patients above and 8 of 22 below the relaxin median died or were readmitted for heart failure (P=.91).
Characteristics of the Total Population and According to Relaxin Concentrations Upon Arrival at the Emergency Room Below or Above the Median
| Total population, n=43 | Relaxin<14.3 (n=22) | Relaxin>14.3 (n=21) | P | |
|---|---|---|---|---|
| Age, y | 69.8±9.4 | 70.6±9.7 | 68.8±9.02 | .518a |
| Sex, female | 16 (37.2) | 10 (45.5) | 5 (28.6) | .258b |
| BMI | 28.9±4.5 | 29.1±4.5 | 28.7±4.6 | .819a |
| HT | 31 (72.1) | 16 (72.7) | 15 (71.4) | .925b |
| Diabetes mellitus | 18 (41.9) | 11 (50) | 7 (33.3) | .274b |
| Dyslipidemia | 16 (38.1) | 8 (36.4) | 8 (40) | .811b |
| History of stroke | 6 (14) | 5 (22.7) | 1 (4.8) | .093b |
| Previous NYHA | 2 (1-2.2) | 2 (1-2.7) | 2 (1-2.2) | .473c |
| Hospitalizations for previous HFs | 15 (37.5) | 5 (25) | 10 (50) | .107b |
| Ischemic heart disease | 14 (32.6) | 7 (31.8) | 7 (33.3) | .917b |
| AMI | 6 (14) | 3 (13.6) | 3 (14.3) | .952b |
| Revascularization | 6 (14) | 1 (4.5) | 5 (23.8) | .072b |
| ICD or CRT user | 1 (2.3) | 0 (0) | 1 (4.8) | .306b |
| APE | 20 (46.5) | 8 (36.4) | 12 (57.1) | .177b |
| SBP, mmHg | 138.9±33.4 | 135±29 | 142±37.5 | .494a |
| DBP, mmHg | 77±17.4 | 75±12.9 | 78.7±21.2 | .527a |
| HR, bpm | 88±27.4 | 88±23 | 87±31 | .937a |
| NMV | 7 (16.7) | 2 (9.5) | 5 (23.8) | .220b |
| Dopamine IV | 3 (7) | 2 (9.1) | 1 (4.8) | .582b |
| Dobutamine IV | 1 (2.3) | 0 (0) | 1 (4.8) | .306b |
| Nitrates IV | 10 (24.4) | 4 (19) | 6 (30) | .420b |
| Sinus rhythm | 22 (51.2) | 11 (50) | 11 (52.4) | .877b |
| Creatinine, mg/dL | 1.1 (0.9-1.4) | 1 (0.89-1.3) | 1.1 (0.95-1.43) | .381c |
| Urea, mg/dL | 45 (39-57) | 45 (37-55) | 49.5 (4-67.2) | .433c |
| Cystatin C, mg/L | 1.37 (1.2-1.9) | 1.53 (1.12-2.27) | 1.33 (1.26-1.91) | .744c |
| Sodium, mEq/L | 140 (137-142) | 139 (135-142) | 140 (137-141) | .855c |
| Potassium, mEq/L | 4.3±0.5 | 4.2±0.5 | 4.3±0.4 | .780a |
| Hemoglobin, g/dL | 12.8±2 | 12.5±1.9 | 13±2 | .454a |
| NT-proBNP, pg/mL | 3932 (172-8149) | 3609 (1619-8152) | 4573 (1726-5600) | .911c |
| Troponin T, pg/mL | 25 (16-46.3) | 23 (15.5-32) | 32.2 (16.1-49.8) | .338c |
| LVEF, % | 40.3±17 | 40.6±17.9 | 39.8±16.3 | .890a |
| LV mass, g/m2 | 131.9±47.5 | 119±43.8 | 144±48.9 | .112a |
| LVEDV, mL/m2 | 70.6±29.1 | 67.8±25.3 | 73.5±33.24 | .567a |
| LVEDD, mm/m2 | 52±11.2 | 51±8.4 | 53±13.5 | .596a |
| SPAP, mmHg | 44.8±13.8 | 48.7±16 | 40±9.2 | .128a |
| LA, mm | 45.5±8.44 | 45.6±6.5 | 45.5±10 | .959a |
AMI, acute myocardial infarction; APE, acute pulmonary edema; BMI, body mass index; CRT, cardiac resynchronization therapy; DBP, diastolic blood pressure; HF, heart failure; HR, heart rate; HT, hypertension; ICD, implantable cardioverter defibrillator; IV, intravenous; LA, anteroposterior diameter of left atrium; LV, left ventricle; LVEDD, left ventricular end-diastolic diameter; LVEDV, left ventricular end-diastolic volume; LVEF, left ventricular ejection fraction; NMV, noninvasive mechanical ventilation; NT-proBNP, N-terminal pro-brain natriuretic peptide; NYHA: New York Heart Association functional class; SBP, systolic blood pressure; SPAP, systolic pulmonary artery pressure.
Our study is the first to investigate relaxin kinetics in patients with AHF and its relationship with a broad set of variables related to the disease. Relaxin concentrations showed considerable variability, as observed in the few published studies, with variations between 600 and 900 (pg/mL)4–6, similar to those of our population (878-1031 pg/mL). These studies included patients with chronic heart failure, with reduced5,6 or preserved6 left ventricular ejection fraction, and after discharge following an episode of AHF4. The lack of an association with clinical variables plus the fact that hormone concentrations do not depend on the time point, whether the patient is in the emergency room with AHF or stabilized at 30 days, indicates that relaxin, as an endogenous hormone, does not actively participate in the pathophysiology of AHF. Once the patient has arrived at the emergency room, sample collection at different time points should have no influence, given the correlation with 30-day fasting values and the lack of a circadian effect on relaxin in previous studies. The results of our study are consistent with the those of series that found similar relaxin concentrations in patients with chronic heart failure and healthy controls5,6 and found no relationship with N-terminal pro-brain natriuretic peptide or prognosis at discharge following an AHF episode4. Only 1 study has observed this relationship, finding that concentrations correlated with ventricular filling pressures and cardiac index and dropped after 48 hours of intravenous nitroprusside in 14 patients with AHF and reduced left ventricular ejection fraction2; however, the decrease was not observed in 13 patients with moderate heart failure. This discrepancy in the findings does not seem to be explained by objective data, such as the type of patient or the clinical picture.
In conclusion, our study shows that relaxin can be detected in the peripheral blood of patients with AHF on arrival at the emergency room, but that relaxin concentrations were highly variable, did not correlate with clinical variables, and showed no significant change after clinical improvement (30 days). These data, along with other publications, indicate that relaxin, as an endogenously produced hormone, has no compensatory role in the pathophysiology of AHF. However, because only a few studies with small numbers of patients have been published, new studies should be conducted to confirm the role of relaxin as a biomarker in heart failure.
FUNDINGThis work was funded by the national Cardiovascular Research Network (Red Nacional de Investigación Cardiovascular [RIC]), Ministry of Health and Consumer Affairs, Madrid, Spain [RD12/0042/0049].