Cardiopulmonary rehabilitation (CR) in patients who have undergone surgery for congenital heart defects (CHDs) is rarely undertaken in Spain, despite its beneficial effects and the fact that physical activity is recommended for CHDs by the European scientific societies.1
An interventional, experimental, prospective, phase I study was conducted (with no randomization for rehabilitation program assignment) to evaluate program safety and functional improvement in 24 young patients (median age, 19 [range, 9-31] years) with complex CHDs that had been treated surgically. This phase 1 study was designed with safety as its primary endpoint and avoided the need to calculate the sample size. The intervention consisted of a 3-month program of twice-weekly CR sessions in groups of 4 or 5 individuals. Each 1-hour session included personalized exercise consisting of warmups, respiratory physiotherapy, aerobic exercise (treadmill, bicycle, and/or videogames), cooldowns, and stretches. Assessments and monitoring were performed in a session with a cardiologist, a physical therapist, a rehabilitation therapist, a psychologist, and a nurse. The program incorporated health instruction, nutritional support, and psychological orientation, with family participation. In addition to ultrasound and electrocardiography, patient assessment included forced spirometry, 6minute walk test, ergospirometry, and quality of life surveys2,3 before and after the program. Patients were not enrolled if they had syndromal CHDs or major comorbidities that could affect or influence the parameters assessed. All patients signed an informed consent form.
Categorical variables are shown as percentages, and continuous variables are shown as the median (range). Nonparametric tests were used to compare dependent paired proportions (McNemar) or ordinal variables (Wilcoxon). A P value <.005 was considered significant.
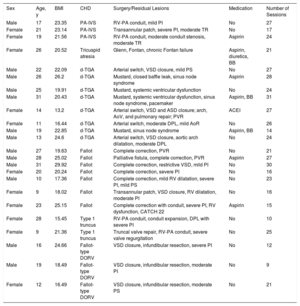
The patient sample is described in table 1. The number of scheduled sessions was 24, with a median adherence of 23.5 (range, 9-31). Patient #18 was treated by pulmonary valve replacement, whereas the others required no therapeutic or medical intervention of any kind. No adverse cardiovascular events or electrocardiographic or echocardiographic changes were reported before or after the program.
Characteristics of patients undergoing the cardiac rehabilitation program
| Sex | Age, y | BMI | CHD | Surgery/Residual Lesions | Medication | Number of Sessions |
|---|---|---|---|---|---|---|
| Male | 17 | 23.35 | PA-IVS | RV-PA conduit, mild PI | No | 27 |
| Female | 21 | 23.14 | PA-IVS | Transannular patch, severe PI, moderate TR | No | 17 |
| Female | 19 | 21.56 | PA-IVS | RV-PA conduit, moderate conduit stenosis, moderate TR | Aspirin | 24 |
| Female | 26 | 20.52 | Tricuspid atresia | Glenn, Fontan, chronic Fontan failure | Aspirin, diuretics, BB | 21 |
| Male | 22 | 22.09 | d-TGA | Arterial switch, VSD closure, mild PS | No | 27 |
| Male | 26 | 26.2 | d-TGA | Mustard, closed baffle leak, sinus node syndrome | Aspirin | 28 |
| Male | 25 | 19.91 | d-TGA | Mustard, systemic ventricular dysfunction | No | 24 |
| Male | 31 | 20.43 | d-TGA | Mustard, systemic ventricular dysfunction, sinus node syndrome, pacemaker | Aspirin, BB | 31 |
| Female | 14 | 13.2 | d-TGA | Arterial switch, VSD and ASD closure; arch, AoV, and pulmonary repair; PVR | ACEI | 27 |
| Female | 11 | 16.44 | d-TGA | Arterial switch, moderate DPL, mild AoR | No | 26 |
| Male | 19 | 22.85 | d-TGA | Mustard, sinus node syndrome | Aspirin, BB | 14 |
| Male | 13 | 24.6 | d-TGA | Arterial switch, VSD closure, aortic arch dilatation, moderate DPL | No | 24 |
| Male | 27 | 19.63 | Fallot | Complete correction, PVR | No | 21 |
| Male | 28 | 25.02 | Fallot | Palliative fistula, complete correction, PVR | Aspirin | 27 |
| Male | 31 | 29.92 | Fallot | Complete correction, restrictive VSD, mild PI | No | 30 |
| Female | 20 | 20.24 | Fallot | Complete correction, severe PI | No | 16 |
| Male | 10 | 17.36 | Fallot | Complete correction, mild RV dilatation, severe PI, mild PS | No | 23 |
| Female | 9 | 18.02 | Fallot | Transannular patch, VSD closure, RV dilatation, moderate PI | No | 16 |
| Female | 23 | 25.15 | Fallot | Complete correction with conduit, severe PI, RV dysfunction, CATCH 22 | Aspirin | 15 |
| Female | 28 | 15.45 | Type 1 truncus | RV-PA conduit, conduit expansion, DPL with severe PI | No | 10 |
| Female | 9 | 21.36 | Type 1 truncus | Truncal valve repair, RV-PA conduit, severe valve regurgitation | No | 25 |
| Male | 16 | 24.66 | Fallot-type DORV | VSD closure, infundibular resection, severe PI | No | 12 |
| Male | 19 | 18.49 | Fallot-type DORV | VSD closure, infundibular resection, moderate PI | No | 9 |
| Female | 12 | 16.49 | Fallot-type DORV | VSD closure, infundibular resection, moderate PS | No | 21 |
ACEI, angiotensin-converting enzyme inhibitor; AoR, aortic regurgitation; AoV, aortic valve; ASD, atrial septal defect; BB, beta-blockers; BMI, body mass index; CHD, congenital heart defect; DORV, double-outlet right ventricle; d-TGA, dextro-transposition of the great arteries; DPL, double pulmonary lesion; PA-IVS, pulmonary atresia with intact ventricular septum; PI, pulmonary insufficiency; PS, pulmonary stenosis; PVR, pulmonary valve replacement; RV, right ventricle; RV-PA, right ventricle-pulmonary artery; TR, tricuspid regurgitation; VSD, ventricular septal defect.
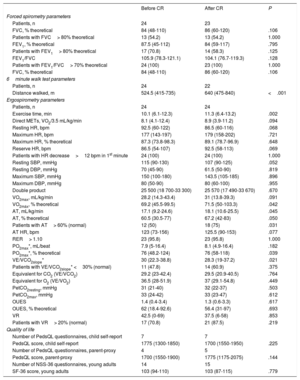
The course of the various parameters assessed before and after CR is shown in table 2. Upon completion of the program, the most significant cardiopulmonary changes were: a) increased inspiratory muscle strength and increased maximal inspiratory pressure; b) greater exertional capacity and tolerance to exercise, with increase in distance walked in the 6-minute walk test; longer exertion time (more than 1minute) and tendency toward better heart rate recovery in the first minute after exertion, as a possible improvement in autonomic nervous system regulation; c) improvement in maximal aerobic capacity, with a significant increase in peak O2 uptake (VO2, expressed as % theoretical); d) improvement in aerobic physical performance, considered a higher VO2 in the anaerobic threshold; e) improvement in cardiocirculatory response, as shown by the lower resting heart rate (with no drug-induced changes), increase in predicted maximal VO2 as an indirect estimator of cardiac output, and in predicted O2 pulse as a parameter to estimate systolic volume at maximal exertion; f) improvement in ventilatory efficiency in exercise, with a decrease in the slope of the plot line for ventilation per minute and CO2 production (VE/VCO2 slope), with a higher number of patients showing a ratio <30, considered normal for patient age and sex. Furthermore, these improvements were achieved in the absence of other changes in ventilatory efficiency and ventricular function variables, as shown by similar values for respiratory equivalents (VE/VCO2, VE/VO2), end-tidal partial pressure of CO2, slope of VO2 efficiency, ventilatory reserve, and echocardiographic measurements of ventricular function before and after the program. These data were consistent with subjective assessments of the New York Heart Association functional class, which reported 18 patients in class I (75%) and 6 in class II (25%) at baseline. By completion of the program, functional class had improved in 4 patients and worsened in 2, for a total of 20 patients in class I (83.3%) and 4 (16.7%) in class II. Last, quality of life questionnaire scores were normal, regardless of the grade of CHD complexity, with no differences between baseline status and the end of the program. The usefulness of the program was highly rated by patients and their families.
Parameters assessed in forced spirometry, 6minute walk test, and ergospirometry, before and after the cardiopulmonary rehabilitation program
| Before CR | After CR | P | |
|---|---|---|---|
| Forced spirometry parameters | |||
| Patients, n | 24 | 23 | |
| FVC, % theoretical | 84 (48-110) | 86 (60-120) | .106 |
| Patients with FVC> 80% theoretical | 13 (54.2) | 13 (54.2) | 1.000 |
| FEV1, % theoretical | 87.5 (45-112) | 84 (59-117) | .795 |
| Patients with FEV1> 80% theoretical | 17 (70.8) | 14 (58.3) | .125 |
| FEV1/FVC | 105.9 (78.3-121.1) | 104.1 (76.7-119.3) | .128 |
| Patients with FEV1/FVC> 70% theoretical | 24 (100) | 23 (100) | 1.000 |
| FVC, % theoretical | 84 (48-110) | 86 (60-120) | .106 |
| 6minute walk test parameters | |||
| Patients, n | 24 | 22 | |
| Distance walked, m | 524.5 (415-735) | 640 (475-840) | <.001 |
| Ergospirometry parameters | |||
| Patients, n | 24 | 24 | |
| Exercise time, min | 10.1 (6.1-12.3) | 11.3 (6.4-13.2) | .002 |
| Direct METs, VO2/3.5 mL/kg/min | 8.1 (4.1-12.4) | 8.9 (3.9-11.2) | .094 |
| Resting HR, bpm | 92.5 (60-122) | 86.5 (60-116) | .068 |
| Maximum HR, bpm | 177 (143-197) | 179 (158-202) | .721 |
| Maximum HR, % theoretical | 87.3 (73.8-98.3) | 89.1 (78.7-96.9) | .648 |
| Reserve HR, bpm | 86.5 (54-107) | 92.5 (58-113) | .069 |
| Patients with HR decrease>12 bpm in 1st minute | 24 (100) | 24 (100) | 1.000 |
| Resting SBP, mmHg | 115 (90-130) | 107 (90-125) | .052 |
| Resting DBP, mmHg | 70 (45-90) | 61.5 (50-90) | .819 |
| Maximum SBP, mmHg | 150 (100-180) | 143.5 (105-185) | .896 |
| Maximum DBP, mmHg | 80 (50-90) | 80 (60-100) | .955 |
| Double product | 25 500 (18 700-33 300) | 25 570 (17 490-33 670) | .670 |
| VO2max, mL/kg/min | 28.2 (14.3-43.4) | 31 (13.8-39.3) | .091 |
| VO2máx, % theoretical | 69.2 (45.5-99.5) | 71.5 (50-103.3) | .042 |
| AT, mL/kg/min | 17.1 (9.2-24.6) | 18.1 (10.6-25.5) | .045 |
| AT, % theoretical | 60.5 (30.5-77) | 67.2 (42-83) | .050 |
| Patients with AT> 60% (normal) | 12 (50) | 18 (75) | .031 |
| AT HR, bpm | 123 (73-156) | 125.5 (90-153) | .077 |
| RER> 1.10 | 23 (95.8) | 23 (95.8) | 1.000 |
| PO2max*, mL/beat | 7.9 (5-16.4) | 8.1 (4.9-16.4) | .182 |
| PO2max*, % theoretical | 76 (48.2-124) | 76 (58-118) | .039 |
| VE/VCO2slope* | 30 (22.3-38.8) | 28.3 (19-37.2) | .021 |
| Patients with VE/VCO2slope* <30% (normal) | 11 (47.8) | 14 (60.9) | .375 |
| Equivalent for CO2 (VE/VCO2) | 29.2 (23-42.4) | 29.5 (20.9-40.5) | .764 |
| Equivalent for O2 (VE/VO2) | 36.5 (28-51.9) | 37 (29.1-54.8) | .449 |
| PetCO2resting, mmHg | 31 (21-40) | 32 (22-37) | .503 |
| PetCO2max, mmHg | 33 (24-42) | 33 (23-47) | .612 |
| OUES | 1.4 (0.4-3.4) | 1.3 (0.6-3.3) | .617 |
| OUES, % theoretical | 62 (18.4-92.6) | 56.4 (31-97) | .693 |
| VR | 42.5 (0-69) | 37.5 (6-58) | .853 |
| Patients with VR> 20% (normal) | 17 (70.8) | 21 (87.5) | .219 |
| Quality of life | |||
| Number of PedsQL questionnaires, child self-report | 7 | 7 | |
| PedsQL score, child self-report | 1775 (1300-1850) | 1700 (1550-1950) | .225 |
| Number of PedsQL questionnaires, parent-proxy | 4 | 5 | |
| PedsQL score, parent-proxy | 1700 (1550-1900) | 1775 (1175-2075) | .144 |
| Number of NSS-36 questionnaires, young adults | 14 | 15 | |
| SF-36 score, young adults | 103 (94-110) | 103 (87-115) | .779 |
AT, anaerobic threshold; CR, cardiopulmonary rehabilitation; DBP, diastolic blood pressure; FEV1, forced expiratory volume in 1 second; FVC, forced vital capacity; HR, heart rate; O2P, oxygen pulse; OUES, oxygen uptake efficiency slope; PedsQL, Pediatric Quality of Life Inventory Cardiac Module, version 4.0 used in our study for pediatric patients (age 8 to 18 years) and their parents; PetCO2, end-tidal partial pressure of CO2; SBP, systolic blood pressure; SF-36, Short Form Health Survey; VCO2, carbon dioxide production; VE, minute ventilation; VO2, oxygen uptake; VR, ventilatory reserve.
Due to medical and surgical advances, it is estimated that more than 85% of children with CHDs in Spain will reach adulthood.4 However, CHD patients who have undergone surgery have lower progressive functional capacity, which increases their morbidity and mortality. In this context, efficient resources for improvement, such as CR, have been implemented; however, they are not widely used in Spain, and there is only 1 published report on experience with 8 patients who had CHDs and pulmonary hypertension,5 with increased functional class and exercise capacity in the 6minute walk test and no adverse events.
The importance of our study is that it is the first to demonstrate the benefits of a CR program in Spain for young people with complex CHDs treated by surgery and that it includes a thorough assessment with ergospirometry. The main limitations of the study are the small, heterogeneous sample and the lack of a control group. Implementation of the program was a challenge, as difficulties were encountered for administration to understand that CR should focus on comprehensive prevention units open to all heart diseases, rather than only coronary patients. We show that, despite these difficulties, CR could be a cost-effective tool capable of improving functional capacity and quality of life in complex CHDs. In our experience, CR has helped to support our patients and their families and enabled them to understand their limits and to encourage improvements in their functional capacity.
FUNDINGBiomedicine, health management, and social and health care research project funded by the Regional Health Agency of Castilla y León (G 1369/A/16) and the CIBERCV, Carlos III Health Institute, Ministry of Science, Innovation, and Universities.