Despite the success of the Spanish heart transplant program, in recent years the number of optimal donors has diminished, leading to longer waiting times and an increase in urgent heart transplantation (HT).1
Patients undergoing urgent transplantation have worse survival and therefore it is recommended that their clinical status be improved by the use of ventricular assist devices. Currently, 15% of patients in Spain are bridged to HT with ventricular assist devices. Because waiting times are below 6 months for an elective HT and less than 1 month for urgent HT,1 most patients can be bridged with short- or mid-term ventricular assist devices. The Berlin Heart EXCOR® (Berlin Heart AG, Berlin, Germany) left ventricular assist devices (LVAD) are a common choice.
We report 2 cases in which an EXCOR® LVAD was replaced by a short-term continuous flow ventricular assist device CentriMag® (Thoratec Corp, Pleasanton, California, United States) as a bridge to transplantation because of thromboembolic complications.
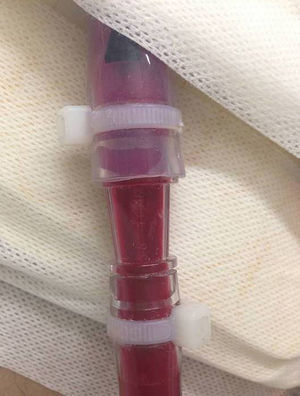
The first case was a 55-year-old man with ischemic dilated cardiomyopathy, who underwent EXCOR® implantation as a bridge to HT candidacy due to irreversible severe pulmonary artery hypertension. Ten days after implantation, while transitioning from heparin to acenocoumarol (international normalized ratio [INR] of 2) and aspirin 100mg, he developed a transient ischemic attack. On day 17, with an INR of 3, a red thrombus was seen in the sinuses of the polyurethane valves (PUVs). Dipyridamole 300mg/d and heparin were added with thrombus resolution. Aggregometry showed arachidonic acid inhibition of 80%. Clopidogrel 75mg/d was added because of persistent white fibrin deposits. On day 40, a right heart catheterization showed reversibility of the pulmonary hypertension. However, in the context of a change to heparin, the patient developed an ischemic stroke. Red thrombi were seen in the sinuses of the PUVs (Figure 1). The EXCOR® LVAD was replaced by a CentriMag® LVAD without resternotomy using the EXCOR® cannulas. One week later, without neurological deficit, the patient was included on the urgent HT list. However, we observed white fibrin deposits in the adaptor that connected the EXCOR® and CentriMag® cannulas (Figure 2). Seventeen days after inclusion on the HT list, the patient developed a catheter-related bacteremia and during transient reduction of heparin for a change of catheter, he developed a new ischemic stroke with hemorrhagic transformation and died.
The second case was a 44-year-old man with ischemic dilated cardiomyopathy who was admitted with decompensated heart failure and needed support with inotropes and intra-aortic balloon pump. The patient was included on the urgent HT list, status 1. Fourteen days later, an EXCOR® LVAD was implanted as a bridge to transplantation. The patient developed cardiac tamponade on the fifth postoperative day, which required the cessation of anticoagulation and surgery. After surgery, heparin and aspirin 100mg/d were restarted but the patient had an ischemic stroke with left hemianopsia and a large red thrombus at the level of the PUV of the outflow cannula. Aggregometry showed no arachidonic acid inhibition and the heart team decided to replace the EXCOR® with a CentriMag® LVAD using the EXCOR® cannulas. Over the next few days, white fibrin deposits were observed in the adaptor connecting the EXCOR® and CentriMag® cannulas, despite treatment with heparin and clopidogrel, but no new neurological deficit developed, so the patient was included for urgent HT, status 0, and was successfully transplanted 17 days after LVAD change.
The incidence of neurological complications in patients with EXCOR® varies among series.2 Management is complex and neurological status frequently becomes a contraindication for HT. We present 2 cases in which EXCOR® implanted as a bridge to transplantation were replaced with short-term continuous flow devices. The reasons for this substitution and for not using another EXCOR® were the following:
- 1.
Thromboembolic complications seemed to be related to a particular component of the EXCOR® LVAD: the PUVs. Until 2014, implanted EXCOR® in adults had mechanical tilting-disk valves. We had no thromboembolic complications in our previous experience. In 2014, we implanted 3 devices with the new PUVs. We suspect that the thrombotic risk in the sinuses of PUVs of the EXCOR® for adults is higher than with the previous mechanical valves, as any reduction of anticoagulation was followed by thromboembolic events. In addition, adults with ischemic cardiomyopathy may have a higher thrombotic risk than pediatric patients, the population in which the safety of these PUVs has been tested.2 The strategy for prevention of thromboembolic complications with the new EXCOR® LVAD is not entirely established. It is likely that more aggressive antiplatelet therapy (by using clopidogrel instead of aspirin) or frequent assessment of platelet function will be recommended for patients with ischemic cardiomyopathy.
- 2.
Waiting times on the urgent HT list, status 0, are still usually below 1 month. The CentriMag® has little thrombogenicity and can give support for up to 1 month, although longer supports have been reported. Because the EXCOR® cannulas can be connected to the CentriMag® LVAD without resternotomy, this strategy is safe in patients with thrombotic complications affecting the extracorporeal circuit of the VAD when the waiting time for urgent HT is short. The only caveat to this approach is that the connection requires an adaptor and we saw thrombus deposition at this level.
In conclusion, we report 2 adult patients in whom we exchanged a long-term LVAD for a short-term LVAD as a bridge to transplantation. We believe the design of the PUVs of the EXCOR® could be partially responsible for the thrombotic complications. A switch to a short-term LVAD is feasible within the Spanish heart transplant program.