Ablation is one of the pillars of treatment for ventricular tachycardias (VT).1,2 Pressure is important for determining the size and potential efficacy of the ablation lesions.3,4 Traditional catheters do not provide direct information on the pressure they exert on tissues; it is estimated indirectly according to the signal voltage and the operator's perceptions. In VT substrate electroanatomical mapping, the absence of contact between the catheter and the endocardium can generate low-voltage electrocardiograms that cause erroneous delimitation of the actual scarring and eventually the loss of the channels involved in maintaining VT. Direct information on the force applied by the tip of the catheter could improve the delimitation of the electrically abnormal tissue in patients with a substrate for VT.
The principal objectives of this study were to compare the scarred areas obtained by traditional voltage mapping with those obtained using a map that includes the contact force information in patients with VT, and to determine whether this information makes it possible to improve the traditional map, correcting the points acquired in areas with no contact.
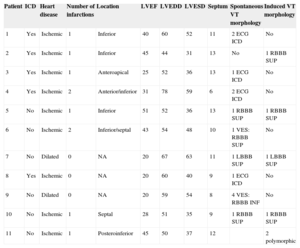
Eleven patients (8 males; mean age, 64.9 [38-80] years) with ventricular arrhythmias due to ischemic cardiomyopathy or dilated cardiomyopathy who underwent VT ablation for clinical indications were included (Table). The study was approved by the Ethics Committee of the hospital and recorded in the clinicaltrials.gov register of clinical trials under identification number NCT01639365.
Demographic and Clinical Characteristics
| Patient | ICD | Heart disease | Number of infarctions | Location | LVEF | LVEDD | LVESD | Septum | Spontaneous VT morphology | Induced VT morphology |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Yes | Ischemic | 1 | Inferior | 40 | 60 | 52 | 11 | 2 ECG ICD | No |
| 2 | Yes | Ischemic | 1 | Inferior | 45 | 44 | 31 | 13 | No | 1 RBBB SUP |
| 3 | Yes | Ischemic | 1 | Anteroapical | 25 | 52 | 36 | 13 | 1 ECG ICD | No |
| 4 | Yes | Ischemic | 2 | Anterior/inferior | 31 | 78 | 59 | 6 | 2 ECG ICD | No |
| 5 | No | Ischemic | 1 | Inferior | 51 | 52 | 36 | 13 | 1 RBBB SUP | 1 RBBB SUP |
| 6 | No | Ischemic | 2 | Inferior/septal | 43 | 54 | 48 | 10 | 1 VES: RBBB SUP | No |
| 7 | No | Dilated | 0 | NA | 20 | 67 | 63 | 11 | 1 LBBB SUP | 1 LBBB SUP |
| 8 | Yes | Ischemic | 0 | NA | 20 | 60 | 40 | 9 | 1 ECG ICD | No |
| 9 | No | Dilated | 0 | NA | 20 | 59 | 54 | 8 | 4 VES: RBBB INF | No |
| 10 | No | Ischemic | 1 | Septal | 28 | 51 | 35 | 9 | 1 RBBB SUP | 1 RBBB SUP |
| 11 | No | Ischemic | 1 | Posteroinferior | 45 | 50 | 37 | 12 | 2 polymorphic |
RBBB, right bundle branch block; LBBB, left bundle branch block; ICD, implantable cardioverter defibrillator; ECG, electrocardiogram; LVEDD, left ventricle end-diastolic diameter; LVEF, left ventricular ejection fraction; LVESD, left ventricle end-systolic diameter; INF, inferior frontal axis; NA, not applicable; SUP, superior frontal axis; VT, ventricular tachycardia; VES, ventricular extrasystole.
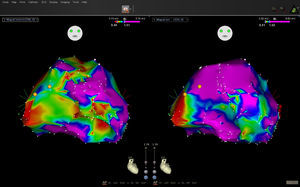
A voltage map of the left ventricle was obtained during sinus rhythm with the Carto® 3 navigation system and a Navistar Thermo Cool catheter (Biosense-Webster; Diamond Bar, California, United States). The points with fragmented or late potentials were labeled, and the color coding of the voltage map was adjusted to locate channels (control map). The information on the force at each point captured was acquired by the system, but was not available to the operator during control-map acquisition. Once this was over, the operator was given the contact information and proceeded to acquire new points in order to correct the previously acquired areas with poor contact (final map). Each point on the control map that had no contact was classified as scarred (< 0.5 mV), low voltage (0.5–1.5 mV), or normal (> 1.5 mV). The size of the scars, the abnormal potentials, and the channels detected in the control map were compared with those of the final map (Figure).
We noticed that in general the mean area did not vary significantly between the control map (10.08 [SD, 6.48] cm2) and the final map (7.69 [SD, 9.21] cm2); however, the area was clearly greater in the initial map of 9 scars and in the final map of 3, and the estimated size was similar in both maps for the remaining 17 scars. The absolute value of the difference in area between maps was 5.38 [SD, 7.32] cm2. In 10 of the 29 scars analyzed, the contact information amended the estimation of the area by more than 50%. Eight conducting channels were detected in the control maps for 6 patients, but were not confirmed in 3 of the patients in the final map. In 3 patients, an additional channel was defined. In 4 patients, areas with low voltage potentials compatible with abnormal areas were observed, which disappeared when the area was remapped in the final map. The areas with no contact in the control maps with potentials < 0.5, 0.5–1.5, and > 1.5mV were 12.3, 18.5, and 8.6cm2, respectively.
The main finding of the study was the demonstration that the use of a catheter with a force sensor makes it possible to define more precisely the true area of scarring as well as the areas with reduced potentials. Our data confirm that a normal voltage does not imply the existence of contact, as the lack of contact can also generate low-voltage electrocardiograms. The detection of channels was also modified by the contact information. The delimitation of channels depends on the difference between the voltages recorded in one area and the adjacent areas. The evidence of no contact in some areas led to more detailed mapping, making it possible to conclude that the values of the potential were more homogeneous and that a false channel had been assumed in the control map. It has been reported that improved contact increases the sensitivity of detection of late potentials5; however, this effect was not observed in our study, possibly due to the small sample size. The choice of the 5g value as an indication of contact was based on the observation that, with the free catheter in the ventricle during system calibration, the force exerted by blood against the catheter sensor was ≤ 5g. Other authors have considered different cut-off values,5 but new studies are needed to define the most appropriate values, both for mapping and for applying radiofrequency with this technology.
We can conclude that inadequate contact of the mapping catheter with the ventricle wall can cause underestimation or overestimation of the extent of scarring on the electroanatomical map and can hinder interpretation of the areas with low potentials, and that the use of an ablation catheter with a force sensor seems to substantially reduce these errors.