Transcatheter aortic valve implantation (TAVI) using a transfemoral (TF) approach is a widely used therapeutic option for the treatment of patients with inoperable severe aortic stenosis or at high surgical risk. Technical improvements and greater operator experience have enhanced the safety of the procedure, although the rate of complications, in particular bleeding and vascular complications, is still high.
Most vascular complications can be rapidly and successfully treated by percutaneous techniques if access is adequate.1 Several authors have described strategies to manage complications via anterograde access from the contralateral femoral2,3 or the radial artery4 or via retrograde access from the ipsilateral femoral artery.5 However, the anterograde techniques described include a delayed approach to therapeutic access at the end of the procedure, when the vascular complication can hinder guidewire introduction and prevent its rapid and adequate management.
The present study evaluated the usefulness of routine placement of a wire in the therapeutic femoral artery from the contralateral femoral artery, introduced at the start of the procedure, to prevent and/or treat vascular complications.
We analyzed data from 159 consecutive patients treated by TF-TAVI at a single hospital between July 2008 and October 2012; in all patients, 18-Fr introducers and percutaneous closure with the Prostar XL device were used after implantation of the Edwards-SAPIEN XT (n=88) or Medtronic CoreValve (n=71) prosthesis.
The patients were divided into 2 sequential groups: group I consisted of patients treated conventionally (no guidewire, n=57, until July 2010), and group II was composed of patients in which a contralateral guidewire was advanced at the start of the procedure (with guide, n=102, as of August 2010). Additionally, in the final 28 patients in this group, a technical variant consisting of balloon inflation at low atmospheres in the puncture area after Prostar closure was used to facilitate hemostasis at this level. The baseline characteristics and 30-day hemorrhagic, vascular, and renal complications according to the Valve Academy Research Consortium (VARC)-2 definitions were compared to analyze therapeutic management in the event of complications.
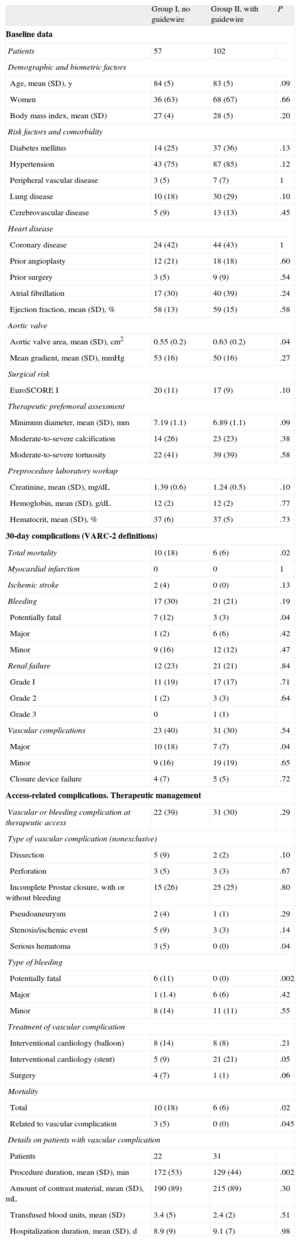
The results are shown in the Table. Both groups had similar baseline characteristics, except for the valvular area, which was larger in group II. Vascular complications and therapeutic femoral bleeding were similar in both groups; the most common complication was incomplete arteriotomy closure with the Prostar device.
Description of Patients’ Baseline Characteristics, Complications, Vascular Complications, and Therapeutic Management
| Group I, no guidewire | Group II, with guidewire | P | |
| Baseline data | |||
| Patients | 57 | 102 | |
| Demographic and biometric factors | |||
| Age, mean (SD), y | 84 (5) | 83 (5) | .09 |
| Women | 36 (63) | 68 (67) | .66 |
| Body mass index, mean (SD) | 27 (4) | 28 (5) | .20 |
| Risk factors and comorbidity | |||
| Diabetes mellitus | 14 (25) | 37 (36) | .13 |
| Hypertension | 43 (75) | 87 (85) | .12 |
| Peripheral vascular disease | 3 (5) | 7 (7) | 1 |
| Lung disease | 10 (18) | 30 (29) | .10 |
| Cerebrovascular disease | 5 (9) | 13 (13) | .45 |
| Heart disease | |||
| Coronary disease | 24 (42) | 44 (43) | 1 |
| Prior angioplasty | 12 (21) | 18 (18) | .60 |
| Prior surgery | 3 (5) | 9 (9) | .54 |
| Atrial fibrillation | 17 (30) | 40 (39) | .24 |
| Ejection fraction, mean (SD), % | 58 (13) | 59 (15) | .58 |
| Aortic valve | |||
| Aortic valve area, mean (SD), cm2 | 0.55 (0.2) | 0.63 (0.2) | .04 |
| Mean gradient, mean (SD), mmHg | 53 (16) | 50 (16) | .27 |
| Surgical risk | |||
| EuroSCORE I | 20 (11) | 17 (9) | .10 |
| Therapeutic prefemoral assessment | |||
| Minimum diameter, mean (SD), mm | 7.19 (1.1) | 6.89 (1.1) | .09 |
| Moderate-to-severe calcification | 14 (26) | 23 (23) | .38 |
| Moderate-to-severe tortuosity | 22 (41) | 39 (39) | .58 |
| Preprocedure laboratory workup | |||
| Creatinine, mean (SD), mg/dL | 1.39 (0.6) | 1.24 (0.5) | .10 |
| Hemoglobin, mean (SD), g/dL | 12 (2) | 12 (2) | .77 |
| Hematocrit, mean (SD), % | 37 (6) | 37 (5) | .73 |
| 30-day complications (VARC-2 definitions) | |||
| Total mortality | 10 (18) | 6 (6) | .02 |
| Myocardial infarction | 0 | 0 | 1 |
| Ischemic stroke | 2 (4) | 0 (0) | .13 |
| Bleeding | 17 (30) | 21 (21) | .19 |
| Potentially fatal | 7 (12) | 3 (3) | .04 |
| Major | 1 (2) | 6 (6) | .42 |
| Minor | 9 (16) | 12 (12) | .47 |
| Renal failure | 12 (23) | 21 (21) | .84 |
| Grade I | 11 (19) | 17 (17) | .71 |
| Grade 2 | 1 (2) | 3 (3) | .64 |
| Grade 3 | 0 | 1 (1) | |
| Vascular complications | 23 (40) | 31 (30) | .54 |
| Major | 10 (18) | 7 (7) | .04 |
| Minor | 9 (16) | 19 (19) | .65 |
| Closure device failure | 4 (7) | 5 (5) | .72 |
| Access-related complications. Therapeutic management | |||
| Vascular or bleeding complication at therapeutic access | 22 (39) | 31 (30) | .29 |
| Type of vascular complication (nonexclusive) | |||
| Dissection | 5 (9) | 2 (2) | .10 |
| Perforation | 3 (5) | 3 (3) | .67 |
| Incomplete Prostar closure, with or without bleeding | 15 (26) | 25 (25) | .80 |
| Pseudoaneurysm | 2 (4) | 1 (1) | .29 |
| Stenosis/ischemic event | 5 (9) | 3 (3) | .14 |
| Serious hematoma | 3 (5) | 0 (0) | .04 |
| Type of bleeding | |||
| Potentially fatal | 6 (11) | 0 (0) | .002 |
| Major | 1 (1.4) | 6 (6) | .42 |
| Minor | 8 (14) | 11 (11) | .55 |
| Treatment of vascular complication | |||
| Interventional cardiology (balloon) | 8 (14) | 8 (8) | .21 |
| Interventional cardiology (stent) | 5 (9) | 21 (21) | .05 |
| Surgery | 4 (7) | 1 (1) | .06 |
| Mortality | |||
| Total | 10 (18) | 6 (6) | .02 |
| Related to vascular complication | 3 (5) | 0 (0) | .045 |
| Details on patients with vascular complication | |||
| Patients | 22 | 31 | |
| Procedure duration, mean (SD), min | 172 (53) | 129 (44) | .002 |
| Amount of contrast material, mean (SD), mL | 190 (89) | 215 (89) | .30 |
| Transfused blood units, mean (SD) | 3.4 (5) | 2.4 (2) | .51 |
| Hospitalization duration, mean (SD), d | 8.9 (9) | 9.1 (7) | .98 |
SD, standard deviation; VARC-2, Valve Academic Research Consortium.
Unless otherwise indicated, the data are expressed as No. (%).
Although there were no differences in the total number of vascular complications, group II showed significant reductions in serious complications, total mortality, and vascular access complications.
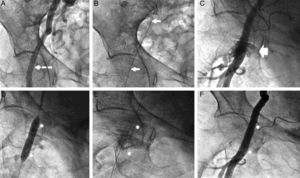
In the therapeutic management of complications, group I had more surgical repairs and more stents than group II (the Figure shows an example of the second group). Interventional cardiology was highly effective in the treatment of vascular complications in both groups, and bleeding was completely resolved in the 26 patients treated with a stent.
Group II patient treated by stent implantation. A: Right femoral angiography; puncture site selection (arrow). B: Angioplasty guidewire (arrows) advanced from the contralateral femoral artery. C: Angiography after Prostar closure; capillary leak syndrome of contrast material is observed in the puncture area. D: Implantation of Advanta V12 drug-eluting stent in the capillary leak syndrome area. E: Fluoroscopic image of the stent (asterisk). F: Final angiographic image.
In patients managed by surgery, the procedure was performed immediately for incontrollable bleeding in 4 patients in group I, and for ischemia at 48 hours postprocedure in the only patient who underwent surgery in group II.
The use of a contralateral guidewire in TF-TAVI procedures was initially described by Sharp et al.2 These authors observed a surgical repair rate of 70% for vascular complications, and reported that balloon compression hemostasis was able to stabilize the patient until the procedure. Buchanan et al4 proposed anterograde left radial access using a long coaxial balloon to promote hemostasis during suturing. If there was persistent bleeding, additional contralateral femoral access was used to attempt percutaneous repair of the complication.
Frerker et al5 described ipsilateral retrograde access and achieved percutaneous repair of most vascular complications with this technique, although with a higher number of minor complications related to dual ipsilateral access.
In our series, the wire was passed from the contralateral femoral artery rapidly and readily in all patients, was not associated with any complications, and allowed immediate percutaneous repair of the complication in all patients.
The use of a contralateral guidewire in TF-TAVI procedures does not decrease the incidence or type of complications, but could reduce the severity and clinical repercussions of these complications and facilitate percutaneous treatment. Because this study was sequential, experience may have led to better outcomes in the second group.