Dextro-transposition of the great arteries (DTGA) is a congenital heart defect characterized by ventriculoarterial discordance, in which the aorta originates at the right ventricle and the pulmonary artery at the left ventricle. The prevalence of this condition is estimated to be between 4 and 6 per 10 000 live newborns, accounting for approximately 20% of all cyanotic congenital heart diseases.1,2 Corrective surgical techniques have advanced over the years from intra-atrial correction (Senning and Mustard) to arterial switching (Jatene), with this latter technique being the technique of choice today.3–5 One of the most common complications of intra-atrial correction is stenosis where the venae cavae enter the right neoatrium.6
We present the case of a 32-year-old woman with DTGA and an intact septum. The Blalock-Hanlon and Blalock-Taussig procedures were performed in the neonatal period, with subsequent anatomical correction with the Mustard technique at the age of 2 years. In subsequent follow-up, sinus dysfunction was observed and, in recent months, the patient complained of dyspnea on moderate effort with reduced functional capacity and desaturation during exercise stress testing.
Cardiac magnetic resonance showed significant stenosis in the superior vena cava (SVC) and the inferior vena cava (IVC), as well as substantial collateral circulation with a very dilated azygos vein. Catheterization revealed moderate stenosis of the SVC (with a mean gradient of 3-mmHg between the right neoatrium and the SVC), complete occlusion of the IVC, and bilateral iliofemoral venous thrombosis. During the catheterization procedure, a 45-mm Cheatham Platinum (CP) stent was implanted over a 22-mm Balloon intraballoon (BIB) in the stenotic region of the SVC with disappearance of the gradient and improvement of the anterograde flow. After catheterization, the patient had an episode of atrial flutter that was reverted with amiodarone. Abdominal echocardiography showed heterogeneity of the hepatic parenchyma. On discussion of the case, it was decided to attempt percutaneous recanalization of the complete occlusion of the IVC.
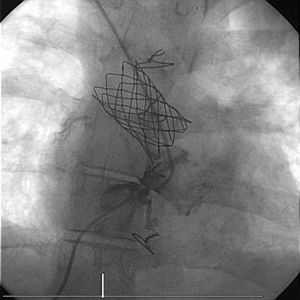
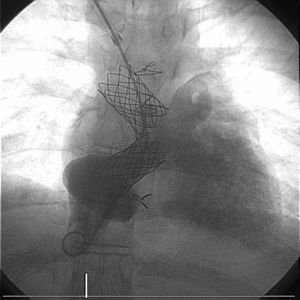
A new interventional procedure was scheduled with the collaboration of the interventional vascular radiology department for transhepatic access to the IVC (5F introducer). The right neoatrium was probed from the right jugular vein and the catheters introduced via the 2 venous accesses (jugular and transhepatic) were placed on either side the occlusion with biplanar fluoroscope (Figure 1). A 0.0014 Nykanen radiofrequency wire was used to perforate the atretic segment from the IVC and captured with a loop catheter to form a venovenous loop. Progressive dilatation of the stenotic region was performed with balloon catheters (Evercross 5, 9, and 12mm diameter) before subsequent 45-mm CP stent implantation over 18-mm BIB with a 12F sheaf introduced via the right jugular vein. The final angiography showed that the caliber of both stents was satisfactory with good flow (Figure 2). Hemostasis was performed by compression. The fluoroscopy time was 52.8minutes. The patient was discharged after 48hours with no complications and her functional class improved during follow-up.
Vascular complications in patients with DTGA who have undergone the Mustard correction are very common and potentially serious. This case illustrates an exceptional situation, that is, complete occlusion of the IVC with no possibility of venous access through the femoral veins, along with use of uncommon techniques for resolving such problems: use of radiofrequency guidewires and transhepatic venous access.
In the follow-up of patients with corrected congenital heart disease, diagnostic imaging tests (magnetic resonance imaging and computed tomography) provide valuable additional information to that provided by echocardiography. Between September 2006 and February 2016, 40 interventional procedures were performed in our center on 35 patients with DTGA who had undergone the Mustard technique. In 10 of these, complete occlusion was observed in the SVC or IVC (n = 7 and n = 2, respectively). Although perforation with a rigid 0.14” guidewire was successful in 7 of 10 patients, we believe that the use of radiofrequency guidewire can increase the success rate in percutaneous recanalization of venous vessels. The use of diagnostic imaging tests (such as magnetic resonance imaging) in the follow-up of these patients can help in the early detection of stenosis of venous vessels.
Collaboration between multidisciplinary teams, use of transhepatic access, biplanar fluoroscopy, and use of perforation techniques with radiofrequency (designed for use in neonatal heart disease), although not authorized in this indication, can be extremely useful in the most complex cases.