To the Editor,
Because of its known safety and efficacy, amiodarone is a first-line drug for the treatment of atrial and ventricular arrhythmias in patients with structural heart disease. Its proarrhythmic side effects are usually underestimated because they are less common than those experienced with other antiarrhythmic drugs. We describe a case of corrected QT interval prolongation (QTc) and torsade de pointes (TdP) secondary to amiodarone.
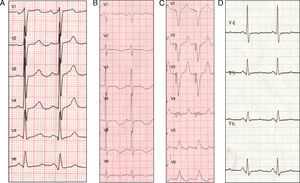
A 59-year-old woman with hypertension and chronic obstructive pulmonary disease with a significant bronchospastic component was attended at home for palpitations and dizziness. The electrocardiogram (ECG) showed self-limiting, regular, wide-QRS tachycardia at a rate of 210 bpm before admission and sinus rhythm at a rate of 90bpm with a QRS duration of 90ms and QTc of 415ms on hospital admission (Figure 1A). The cardiologic study revealed dilated cardiomyopathy with moderate ventricular dysfunction, but no major coronary lesions.
Figure 1. A, baseline electrocardiogram during sinus rhythm (90bpm, QRS 90ms, corrected QT interval 415ms). B, electrocradiogram during sinus rhythm 24h post-amiodarone dosing (75bpm, QRS 146ms, corrected QT interval 714ms). C, pacemaker with pacing at 100bpm. D, electrocardiogram during sinus rhythm 1 week after amiodarone discontinuation (75bpm, QRS 110ms, corrected QT interval 449ms).
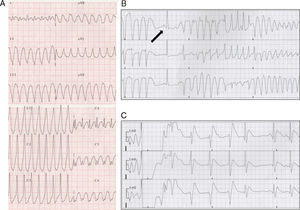
On the third day of hospitalization, the patient experienced well-tolerated sustained monomorphic ventricular tachycardia (SMVT) at a rate of 270bpm (Figure 2A) that reverted once intravenous perfusion of amiodarone was initiated.
Figure 2. A, sustained monomorphic ventricular tachycardia. B, torsade de pointes. Note the sinus beat with long corrected QT interval preceding it (arrow). C, electrical cardioversion of ventricular fibrillation.
Twenty-four hours later, the patient presented an episode of TdP that degenerated into ventricular fibrillation, which was treated by electrical cardioversion (Figure 2B and C), and therefore she was transferred to our hospital.
The ECG on admission showed sinus rhythm at a rate of 75bpm with a QRS duration of 146ms and QTc of 714ms (Figure 1B). The patient presented no other recognizable factors that prolong QTc interval, except discrete hypopotassemia (3.2mEq/L).
Although amiodarone was discontinued and potassium and magnesium supplements were given, she experienced 11 new episodes of TdP, which were controlled by implantation of a temporary ventricular pacemaker at 100bpm (Figure 1C) for 4 days. A week after amiodarone discontinuation, the QRS measured 110ms and the QTc, 449ms (Figure 1D).
In view of the well tolerated SMVT episodes and the moderate nonischemic ventricular dysfunction, an electrophysiologic study was performed in which clinical SMVT originating in the apical lateral region of the left ventricle was induced and the circuit was ablated.
Amiodarone acts by blocking different ion channels involved in the action potential, which makes it effective for the treatment of various arrhythmias. The drug's dominant effect is class III, as it blocks potassium channels during repolarization and therefore can prolong the QTc interval.
QTc elongation is known to be the predisposing factor for the appearance of TdP, and although amiodarone can prolong the QTc few reports of amiodarone-induced TdP have been published.
Several small series found no correlation between amiodarone-induced QTc prolongation and the onset of TdP, which indicates that it is safe even in patients with this arrhythmic complication.1
The association was also detected in experimental animal studies, from which it can be inferred that the absence of TdP with amiodarone may be related to homogeneous lengthening of the action potential duration and the absence of early afterdepolarizations.2
Although the incidence of TdP with amiodarone is low compared to other class III antiarrhythmic agents, it is known to be twice as high in women.3, 4 Some published case reports have detected other associated factors that could prolong QTc (electrolyte abnormalities, bradycardia, etc.), which could enhance the proarrhythmic effect of the drug.5
In our patient, QTc prolongation showed a clear temporal relationship with amiodarone dosing both in terms of onset (24h afterwards) and disappearance (1 week after the drug was discontinued).
Although slight hypopotassemia was observed, TdP did not cease when the levels were corrected, which suggests that although the condition could have enhanced the proarrhythmic effect of the drug it was not the main cause.
In conclusion, although amiodarone is considered safe for the treatment of ventricular arrhythmia, its arrhythmogenic potential should not be underestimated, particularly in women and in the presence of concomitant factors that could prolong the QTc. Careful monitoring of the QTc interval and these factors can lower the risk of proarrhythmia.
.
Corresponding author: alfonjroman@hotmail.com