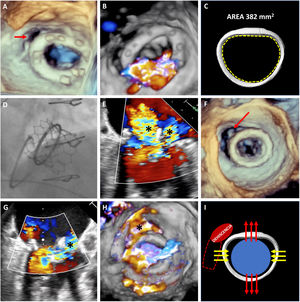
A 73-year-old woman who had undergone mitral and tricuspid annuloplasty with a 28-mm Physio mitral ring and a 34-mm MC3 tricuspid ring (Edwards Lifesciences, USA) developed severe symptomatic mitral regurgitation with minor anterolateral ring dehiscence but no evidence of paravascular leakage (PVL) (figure 1A,B, arrow, and ). Because of the patient's comorbidities, it was decided to perform percutaneous mitral valve-in-ring implantation. The internal area of the ring estimated by computed tomography (CT) was 382mm2 (figure 1C), which was compatible with a 23-mm Edwards SAPIEN-3 valve. The Mitral Valve-in-Valve (ViV) application, however, recommended a 26-mm ring (figure 1D). The larger ring was eventually used but resulted in intraprosthetic regurgitation, due to incomplete closure of the leaflets, and periprosthetic regurgitation (figure 1E, asterisks, and ). The intraprosthetic regurgitation was corrected with a 28-mm balloon, but this caused a considerable increase in the size of the dehiscence and severe PVL (figure 1F-H, arrow and asterisks, and ). The patient underwent surgical repair with a favorable outcome. Percutaneous repair was ruled out because of the size of the defect (> 180°) and because it had probably been caused by annular tear.
The present case illustrates 2 potential complications of mitral valve-in-ring implantation that could have an unfavorable outcome. The difference between the size of the ring observed by CT and that recommended by the ViV application resulted in the use of an oversized ring, which might have altered the mobility of the heart valve leaflets. It will also have increased the tension on an already dehisced ring in a critical area (figure 1I), with worsening of the situation after postdilation. This case highlights the importance of correct sizing and of evaluating different factors that can affect the outcome of mitral valve-in-ring implantation. CT appears to provide the most reliable estimate of ring size, but more studies are needed to confirm this hypothesis. We obtained written informed consent from our patient for the publication of this case.
FundingNo funding was received.
Authors’ ContributionsF. Sabatel-Pérez procured the images, wrote the manuscript, and created and corrected the figure. F. Carrasco-Chinchilla created the figure and revised and corrected the manuscript. JH Alonso-Briales revised and corrected the manuscript and provided scientific counseling.
Conflicts of InterestThe authors declare that they do not have any conflicts of interest in relation to this article.
Supplementary data associated with this article can be found in the online version available at https://doi.org/10.1016/j.recesp.2021.04.005